|
|
市場調査レポート
商品コード
1545608
代用人工血液市場- 世界および地域別分析:用途別、最終用途別、製品別、由来別、タイプ別、構成別、地域別 - 分析と予測(2024年~2034年)Synthetic Blood Substitute Market - A Global and Regional Analysis: Focus on Application, End-use, Product, Source, Type, Configuration, and Region - Analysis and Forecast, 2024-2034 |
||||||
カスタマイズ可能
|
|||||||
| 代用人工血液市場- 世界および地域別分析:用途別、最終用途別、製品別、由来別、タイプ別、構成別、地域別 - 分析と予測(2024年~2034年) |
|
出版日: 2024年09月02日
発行: BIS Research
ページ情報: 英文 105 Pages
納期: 1~5営業日
|
全表示
- 概要
- 目次
代用人工血液の市場規模は、従来の輸血の限界に対処するための重要なソリューションとして急速に台頭しており、世界の様々な医療分野で広く応用されています。
外傷、手術、慢性疾患の増加に伴い、代用人工血液は信頼性が高く安全な代替手段を提供し、重要な血液成分の中断のない供給を保証します。楽観的シナリオでは、2024年に870万米ドルと評価された市場は、CAGR 15.43%で成長し、2034年には3,670万米ドルに達すると予想されています。
医療の進歩と信頼性の高い代替血液へのニーズの高まりが、ヘルスケア分野における代用人工血液の需要を促進しています。外傷、手術、慢性疾患の発生率が上昇を続ける中、従来の輸血の限界を克服できる効果的で安全な代用血液の必要性が高まっています。代用人工血液は、天然血液の酸素運搬機能と凝固機能を模倣する能力を持ち、救急医療、手術、慢性疾患管理などの用途で重要なソリューションとして浮上しています。
| 主要市場統計 | |
|---|---|
| 予測期間 | 2024年~2034年 |
| 2024年の評価 | 870万米ドル |
| 2034年の予測 | 3,670万米ドル |
| CAGR | 15.43% |
代用人工血液市場には、高い研究開発コストや規制当局の承認に伴う複雑さなどの課題があります。さらに、多様な病状におけるこれらの代用血液の安全性と有効性を確保することが、市場への参入と拡大にさらなる複雑さをもたらしています。しかし、このような課題はバイオテクノロジー企業、ヘルスケアプロバイダー、規制機関の間の技術革新と協力を促進し、より高度な代用人工血液の開発を促進し、現代医療におけるその潜在的な用途を拡大しています。
米国を筆頭とする北米は、高度な生物医学研究部門と充実したヘルスケア・インフラに牽引され、代用人工血液市場を独占しています。米国は、研究開発への旺盛な投資と規制機関からの強力な支援により、代用人工血液品開発をリードしています。このような環境が技術革新を促進し、外傷治療、手術、慢性疾患管理における重要なニーズに対応する最先端の合成血液製剤を生み出しています。さらに、米国はヘルスケアの成果を向上させ、血液供給不足に対処することに取り組んでいるため、さまざまな医療分野で代用人工血液の採用が進んでいます。患者の安全性と技術の進歩に重点を置く米国では、代用人工血液の有効性と信頼性が絶えず改良されています。医療を強化し、増加する代用血液の需要に応えることに重点を置く米国は、世界の人工血液市場における主要国としての地位を確固たるものにしています。
当レポートでは、世界の代用人工血液市場について調査し、市場の概要とともに、用途別、最終用途別、製品別、由来別、タイプ別、構成別、地域別の動向、および市場に参入する企業のプロファイルなどを提供しています。
目次
エグゼクティブサマリー
第1章 市場:業界の展望
- 動向:現在および将来の影響評価
- サプライチェーンの概要
- R&Dレビュー
- 規制状況
- ステークホルダー分析
- 主要な世界的イベントの影響分析
- 市場力学の概要
第2章 代用人工血液市場(用途別)
- 用途のセグメンテーション
- 用途の概要
- 代用人工血液市場(用途別)
- 代用人工血液市場(最終用途別)
第3章 代用人工血液市場(製品別)
- 製品セグメンテーション
- 製品概要
- 代用人工血液市場(製品別)
- 代用人工血液市場(由来別)
- 代用人工血液市場(タイプ別)
- 代用人工血液市場(構成別)
第4章 代用人工血液市場(地域別)
- 代用人工血液市場(地域別)
- 北米
- 欧州
- アジア太平洋
- その他の地域
第5章 企業プロファイル
- 今後の見通し
- 地理的評価
- 企業プロファイル
- Aurum Biosciences
- HEMARINA
- Hemoglobin Oxygen Therapeutics LLC
- KaloCyte, Inc.
- Spheritech Ltd
- VisusMed Medical Center
- OPKO Health, Inc.
- NuvOx Pharma
- Prolong Pharmaceuticals
- Boston Pharmaceuticals
- ATPL
- Baxter
- Biopharma corp
- NORTHFIELD LABORATORIES INC.
- Alliance Pharma PLC
- その他の主要企業
第6章 調査手法
Introduction to Synthetic Blood Substitute Market
The synthetic blood substitute market is rapidly emerging as a critical solution to address the limitations of traditional blood transfusions, finding widespread applications across various medical fields globally. With increasing incidences of trauma, surgeries, and chronic diseases, synthetic blood substitutes offer a reliable and safe alternative, ensuring an uninterrupted supply of vital blood components. In an optimistic scenario, the market, valued at $8.7 million in 2024, is anticipated to grow at a CAGR of 15.43%, reaching $36.7 million by 2034.
Increasing medical advancements and a growing need for reliable blood alternatives are driving the demand for synthetic blood substitutes in the healthcare sector. As incidences of trauma, surgeries, and chronic diseases continue to rise, there is a growing necessity for effective and safe blood substitutes that can overcome the limitations of traditional blood transfusions. Synthetic blood substitutes, with their ability to mimic the oxygen-carrying and clotting functions of natural blood, have emerged as a critical solution across applications such as emergency medicine, surgeries, and chronic disease management.
| KEY MARKET STATISTICS | |
|---|---|
| Forecast Period | 2024 - 2034 |
| 2024 Evaluation | $8.7 Million |
| 2034 Forecast | $36.7 Million |
| CAGR | 15.43% |
In the synthetic blood substitute market, challenges include high research and development costs and the complexities associated with regulatory approvals. Additionally, ensuring the safety and efficacy of these substitutes in diverse medical conditions adds further complexity to market entry and expansion. However, these challenges promotes innovation and collaboration among biotech firms, healthcare providers, and regulatory bodies, driving the development of more advanced synthetic blood substitutes and expanding their potential applications in modern medicine.
North America, led by the U.S., dominates the synthetic blood substitute market, driven by its advanced biomedical research sector and substantial healthcare infrastructure. U.S. leads in synthetic blood substitute development due to its robust investment in research and development, coupled with strong support from regulatory bodies. This environment promotes innovation, resulting in cutting-edge synthetic blood products that address critical needs in trauma care, surgeries, and chronic disease management. Furthermore, the U.S. commitment to improving healthcare outcomes and addressing blood supply shortages propels the adoption of synthetic blood substitutes across various medical fields. The nation's emphasis on patient safety and technological advancements ensures that synthetic blood substitutes are continually refined for efficacy and reliability. With a focus on enhancing medical care and meeting the increasing demand for blood substitutes, the U.S. solidifies its position as a key player in the global synthetic blood substitute market.
Key players such as Aurum Biosciences, HEMARINA, Hemoglobin Oxygen Therapeutics LLC, and KaloCyte, Inc. are at the forefront of market expansion, utilizing their technological expertise and strategic partnerships to drive innovation and capture a significant market share. Companies are investing in the construction of new production facilities and the expansion of existing ones to meet the growing demand. For instance, in September 2022, VirTech Bio received approximately $13.7 million in funding from the Pentagon and the U.S. Department of Defense (DoD) to advance the clinical research of its hemoglobin-based oxygen-carrying solution, OxyBridge. This product is designed to prevent hemorrhagic shock in trauma patients and enhance the viability of organs donated for transplantation.
Market Segmentation:
Segmentation 1: by Application
- Cardiovascular Diseases
- Malignant Neoplasma
- Injuries
- Neonatal Conditions
- Organ Transplant
- Maternal Condition
- Others
Segmentation 2: by End-use
- Hospitals
- Ambulatory Surgical Centers
- Military and Defense
- Research Laboratories
- Emergency Services
- Others
Segmentation 3: by Product
- Hemoglobin-Based Oxygen Carriers (HBOCs)
- Perfluorocarbon Emulsions (PFCs)
- Stem Cell-Derived Red Blood Cells
- Others
Segmentation 4: by Source
- Human Blood
- Microorganism Based Recombinant HB
- Synthetic Polymers
- Stem Cells
- Others
Segmentation 5: by Component
- Red Blood Cell Substitutes
- Platelet Substitutes
- Plasma Substitutes
Segmentation 6: by Configuration
- Injectable Solutions
- Powdered Forms
- Infusion Bags
- Capsules and Tablets
- Others
Segmentation 7: by Region
- North America
- Europe
- Asia-Pacific
- Rest-of-the-World
How can this report add value to an organization?
Product/Innovation Strategy: The global synthetic blood substitute market has been extensively segmented based on various categories, such as application, end-use, product, source, component, and configuration. This can help readers get a clear overview of which segments account for the largest share and which ones are well-positioned to grow in the coming years.
Competitive Strategy: A detailed competitive benchmarking of the players operating in the global synthetic blood substitute market has been done to help the reader understand how players stack against each other, presenting a clear market landscape. Additionally, comprehensive competitive strategies such as partnerships, agreements, and collaborations will aid the reader in understanding the untapped revenue pockets in the market.
Key Market Players and Competition Synopsis
The companies that are profiled have been selected based on thorough secondary research, which includes analyzing company coverage, product portfolio, market penetration, and insights gathered from primary experts.
Some of the prominent companies in this market are:
- Aurum Biosciences
- HEMARINA
- Hemoglobin Oxygen Therapeutics LLC
- KaloCyte, Inc.
- Spheritech Ltd
- NuvOx Pharma
- Prolong Pharmaceuticals
- Boston Pharmaceuticals
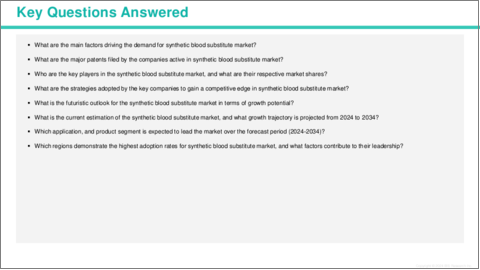
Key Questions Answered in this Report:
- What are the main factors driving the demand for synthetic blood substitute market?
- What are the major patents filed by the companies active in synthetic blood substitute market?
- Who are the key players in the synthetic blood substitute market, and what are their respective market shares?
- What are the strategies adopted by the key companies to gain a competitive edge in synthetic blood substitute market?
- What is the futuristic outlook for the synthetic blood substitute market in terms of growth potential?
- What is the current estimation of the synthetic blood substitute market, and what growth trajectory is projected from 2024 to 2034?
- Which application, and product segment is expected to lead the market over the forecast period (2024-2034)?
- Which regions demonstrate the highest adoption rates for synthetic blood substitute market, and what factors contribute to their leadership?
Table of Contents
Executive Summary
Scope and Definition
Market/Product Definition
Key Questions Answered
Analysis and Forecast Note
1. Markets: Industry Outlook
- 1.1 Trends: Current and Future Impact Assessment
- 1.1.1 Increasing Demand for Blood Transfusions due to Rising Trauma and Chronic Diseases
- 1.1.2 Advancement in Stem Cell Technology and Regenerative Medicine
- 1.1.3 Integration with Advanced Diagnostics and Monitoring
- 1.2 Supply Chain Overview
- 1.2.1 Value Chain Analysis
- 1.2.2 Pricing Forecast
- 1.3 R&D Review
- 1.3.1 Patent Filing Trend by Country, by Company
- 1.4 Regulatory Landscape
- 1.5 Stakeholder Analysis
- 1.5.1 Use Case
- 1.5.2 End User and Buying Criteria
- 1.6 Impact Analysis for Key Global Events
- 1.7 Market Dynamics Overview
- 1.7.1 Market Drivers
- 1.7.2 Market Restraints
- 1.7.3 Market Opportunities
2. Synthetic Blood Substitute Market (by Application)
- 2.1 Application Segmentation
- 2.2 Application Summary
- 2.3 Synthetic Blood Substitute Market (by Application)
- 2.3.1 Cardiovascular Diseases
- 2.3.2 Malignant Neoplasma
- 2.3.3 Injuries
- 2.3.4 Neonatal Conditions
- 2.3.5 Organ Transplant
- 2.3.6 Maternal Condition
- 2.3.7 Others
- 2.4 Synthetic Blood Substitute Market (by End-use)
- 2.4.1 Hospitals
- 2.4.2 Ambulatory Surgical Centers
- 2.4.3 Military and Defense
- 2.4.4 Research Laboratories
- 2.4.5 Emergency Services
- 2.4.6 Others
3. Synthetic Blood Substitute Market (by Product)
- 3.1 Product Segmentation
- 3.2 Product Summary
- 3.3 Synthetic Blood Substitute Market (by Product)
- 3.3.1 Hemoglobin-Based Oxygen Carriers (HBOCs)
- 3.3.2 Perfluorocarbon Emulsions (PFCs)
- 3.3.3 Stem Cell-Derived Red Blood Cells
- 3.3.4 Others
- 3.4 Synthetic Blood Substitute Market (by Source)
- 3.4.1 Human Blood
- 3.4.2 Microorganism Based Recombinant HB
- 3.4.3 Synthetic Polymers
- 3.4.4 Stem Cells
- 3.4.5 Others
- 3.5 Synthetic Blood Substitute Market (by Type)
- 3.5.1 Red Blood Cell Substitutes
- 3.5.2 Platelet Substitutes
- 3.5.3 Plasma Substitutes
- 3.6 Synthetic Blood Substitute Market (by Configuration)
- 3.6.1 Injectable Solutions
- 3.6.2 Powdered Forms
- 3.6.3 Infusion Bags
- 3.6.4 Capsules and Tablets
- 3.6.5 Others
4. Synthetic Blood Substitute Market (by Region)
- 4.1 Synthetic Blood Substitute Market (by Region)
- 4.2 North America
- 4.2.1 Regional Overview
- 4.2.2 Driving Factors for Market Growth
- 4.2.3 Factors Challenging the Market
- 4.2.4 Application
- 4.2.5 Product
- 4.2.6 North America Synthetic Blood Substitute Market (by Country)
- 4.2.6.1 U.S.
- 4.2.6.1.1 Market by Application
- 4.2.6.1.2 Market by Product
- 4.2.6.2 Canada
- 4.2.6.2.1 Market by Application
- 4.2.6.2.2 Market by Product
- 4.2.6.3 Mexico
- 4.2.6.3.1 Market by Application
- 4.2.6.3.2 Market by Product
- 4.2.6.1 U.S.
- 4.3 Europe
- 4.3.1 Regional Overview
- 4.3.2 Driving Factors for Market Growth
- 4.3.3 Factors Challenging the Market
- 4.3.4 Application
- 4.3.5 Product
- 4.3.6 Europe Synthetic Blood Substitute Market (by Country)
- 4.3.6.1 Germany
- 4.3.6.1.1 Market by Application
- 4.3.6.1.2 Market by Product
- 4.3.6.2 France
- 4.3.6.2.1 Market by Application
- 4.3.6.2.2 Market by Product
- 4.3.6.3 U.K.
- 4.3.6.3.1 Market by Application
- 4.3.6.3.2 Market by Product
- 4.3.6.4 Italy
- 4.3.6.4.1 Market by Application
- 4.3.6.4.2 Market by Product
- 4.3.6.5 Rest-of-Europe
- 4.3.6.5.1 Market by Application
- 4.3.6.5.2 Market by Product
- 4.3.6.1 Germany
- 4.4 Asia-Pacific
- 4.4.1 Regional Overview
- 4.4.2 Driving Factors for Market Growth
- 4.4.3 Factors Challenging the Market
- 4.4.4 Application
- 4.4.5 Product
- 4.4.6 Asia-Pacific Synthetic Blood Substitute Market (by Country)
- 4.4.6.1 China
- 4.4.6.1.1 Market by Application
- 4.4.6.1.2 Market by Product
- 4.4.6.2 Japan
- 4.4.6.2.1 Market by Application
- 4.4.6.2.2 Market by Product
- 4.4.6.3 India
- 4.4.6.3.1 Market by Application
- 4.4.6.3.2 Market by Product
- 4.4.6.4 South Korea
- 4.4.6.4.1 Market by Application
- 4.4.6.4.2 Market by Product
- 4.4.6.5 Rest-of-Asia-Pacific
- 4.4.6.5.1 Market by Application
- 4.4.6.5.2 Market by Product
- 4.4.6.1 China
- 4.5 Rest-of-the-World
- 4.5.1 Regional Overview
- 4.5.2 Driving Factors for Market Growth
- 4.5.3 Factors Challenging the Market
- 4.5.4 Application
- 4.5.5 Product
- 4.5.6 Rest-of-the-World Synthetic Blood Substitute Market (by Region)
- 4.5.6.1 South America
- 4.5.6.1.1 Market by Application
- 4.5.6.1.2 Market by Product
- 4.5.6.2 Middle East and Africa
- 4.5.6.2.1 Market by Application
- 4.5.6.2.2 Market by Product
- 4.5.6.1 South America
5. Companies Profiled
- 5.1 Next Frontiers
- 5.2 Geographic Assessment
- 5.3 Company Profiles
- 5.3.1 Aurum Biosciences
- 5.3.1.1 Overview
- 5.3.1.2 Top Products/Product Portfolio
- 5.3.1.3 Top Competitors
- 5.3.1.4 Target Customers
- 5.3.1.5 Key Personnel
- 5.3.1.6 Analyst View
- 5.3.1.7 Market Share
- 5.3.2 HEMARINA
- 5.3.2.1 Overview
- 5.3.2.2 Top Products/Product Portfolio
- 5.3.2.3 Top Competitors
- 5.3.2.4 Target Customers
- 5.3.2.5 Key Personnel
- 5.3.2.6 Analyst View
- 5.3.2.7 Market Share
- 5.3.3 Hemoglobin Oxygen Therapeutics LLC
- 5.3.3.1 Overview
- 5.3.3.2 Top Products/Product Portfolio
- 5.3.3.3 Top Competitors
- 5.3.3.4 Target Customers
- 5.3.3.5 Key Personnel
- 5.3.3.6 Analyst View
- 5.3.3.7 Market Share
- 5.3.4 KaloCyte, Inc.
- 5.3.4.1 Overview
- 5.3.4.2 Top Products/Product Portfolio
- 5.3.4.3 Top Competitors
- 5.3.4.4 Target Customers
- 5.3.4.5 Key Personnel
- 5.3.4.6 Analyst View
- 5.3.4.7 Market Share
- 5.3.5 Spheritech Ltd
- 5.3.5.1 Overview
- 5.3.5.2 Top Products/Product Portfolio
- 5.3.5.3 Top Competitors
- 5.3.5.4 Target Customers
- 5.3.5.5 Key Personnel
- 5.3.5.6 Analyst View
- 5.3.5.7 Market Share
- 5.3.6 VisusMed Medical Center
- 5.3.6.1 Overview
- 5.3.6.2 Top Products/Product Portfolio
- 5.3.6.3 Top Competitors
- 5.3.6.4 Target Customers
- 5.3.6.5 Key Personnel
- 5.3.6.6 Analyst View
- 5.3.6.7 Market Share
- 5.3.7 OPKO Health, Inc.
- 5.3.7.1 Overview
- 5.3.7.2 Top Products/Product Portfolio
- 5.3.7.3 Top Competitors
- 5.3.7.4 Target Customers
- 5.3.7.5 Key Personnel
- 5.3.7.6 Analyst View
- 5.3.7.7 Market Share
- 5.3.8 NuvOx Pharma
- 5.3.8.1 Overview
- 5.3.8.2 Top Products/Product Portfolio
- 5.3.8.3 Top Competitors
- 5.3.8.4 Target Customers
- 5.3.8.5 Key Personnel
- 5.3.8.6 Analyst View
- 5.3.8.7 Market Share
- 5.3.9 Prolong Pharmaceuticals
- 5.3.9.1 Overview
- 5.3.9.2 Top Products/Product Portfolio
- 5.3.9.3 Top Competitors
- 5.3.9.4 Target Customers
- 5.3.9.5 Key Personnel
- 5.3.9.6 Analyst View
- 5.3.9.7 Market Share
- 5.3.10 Boston Pharmaceuticals
- 5.3.10.1 Overview
- 5.3.10.2 Top Products/Product Portfolio
- 5.3.10.3 Top Competitors
- 5.3.10.4 Target Customers
- 5.3.10.5 Key Personnel
- 5.3.10.6 Analyst View
- 5.3.10.7 Market Share
- 5.3.11 ATPL
- 5.3.11.1 Overview
- 5.3.11.2 Top Products/Product Portfolio
- 5.3.11.3 Top Competitors
- 5.3.11.4 Target Customers
- 5.3.11.5 Key Personnel
- 5.3.11.6 Analyst View
- 5.3.11.7 Market Share
- 5.3.12 Baxter
- 5.3.12.1 Overview
- 5.3.12.2 Top Products/Product Portfolio
- 5.3.12.3 Top Competitors
- 5.3.12.4 Target Customers
- 5.3.12.5 Key Personnel
- 5.3.12.6 Analyst View
- 5.3.12.7 Market Share
- 5.3.13 Biopharma corp
- 5.3.13.1 Overview
- 5.3.13.2 Top Products/Product Portfolio
- 5.3.13.3 Top Competitors
- 5.3.13.4 Target Customers
- 5.3.13.5 Key Personnel
- 5.3.13.6 Analyst View
- 5.3.13.7 Market Share
- 5.3.14 NORTHFIELD LABORATORIES INC.
- 5.3.14.1 Overview
- 5.3.14.2 Top Products/Product Portfolio
- 5.3.14.3 Top Competitors
- 5.3.14.4 Target Customers
- 5.3.14.5 Key Personnel
- 5.3.14.6 Analyst View
- 5.3.14.7 Market Share
- 5.3.15 Alliance Pharma PLC
- 5.3.15.1 Overview
- 5.3.15.2 Top Products/Product Portfolio
- 5.3.15.3 Top Competitors
- 5.3.15.4 Target Customers
- 5.3.15.5 Key Personnel
- 5.3.15.6 Analyst View
- 5.3.15.7 Market Share
- 5.3.16 Other Key Companies
- 5.3.1 Aurum Biosciences