|
|
市場調査レポート
商品コード
1364756
臨床試験用品の世界市場 (~2028年):サービス・フェーズ・タイプ・治療領域・エンドユーザー・地域別Clinical Trial Supplies Market by Services (Manufacturing, Packaging, Logistics), Phases, Type (Small Molecules, Biologics), Therapeutic Areas (Oncology, CVD, Infectious, Immunology), End User (Pharma, Biotech, CROs), & Region - Global Forecast to 2028 |
||||||
|
● お客様のご希望に応じて、既存データの加工や未掲載情報(例:国別セグメント)の追加などの対応が可能です。 詳細はお問い合わせください。 |
|||||||
| 臨床試験用品の世界市場 (~2028年):サービス・フェーズ・タイプ・治療領域・エンドユーザー・地域別 |
|
出版日: 2023年10月09日
発行: MarketsandMarkets
ページ情報: 英文 328 Pages
納期: 即納可能
|
- 全表示
- 概要
- 目次
世界の臨床試験用品の市場規模は、2023年の39億米ドルから、予測期間中は9.9%のCAGRで推移し、2028年には63億米ドルの規模に成長すると予測されています。
臨床試験数の増加、製品パイプラインの成長、臨床試験の分散化の進行などの要因が、市場成長にプラスの影響を与えると考えられています。
| 調査範囲 | |
|---|---|
| 調査対象年 | 2021-2028年 |
| 基準年 | 2022年 |
| 予測期間 | 2023-2028年 |
| 単位 | 米ドル |
| セグメント別 | サービス・フェーズ・タイプ・治療領域・エンドユーザー別 |
| 対象地域 | 北米・欧州・アジア太平洋・ラテンアメリカ・中東&アフリカ |
フェーズIIの部門が2022年に最大のシェアを占める
フェーズ別で見ると、フェーズIIの部門が2022年に最大のシェアを示しています。同部門は、臨床試験登録数の増加や、新しい癌治療薬の開発のための製薬・バイオテクノロジー企業によるR&D投資の増加など、さまざまな要因から市場で圧倒的なシェアを示しています。
アジア太平洋地域がより速いペースで成長する可能性が高い
地域別では、アジア太平洋地域が予測期間中に大きなCAGRで成長すると予測されています。同地域の成長の主な原動力は、登録臨床試験数の増加です。中国、インド、日本では、臨床試験サービスを提供する有力企業が存在するため、臨床試験の分散化が進んでおり、アジア太平洋地域の市場成長を支えています。
当レポートでは、世界の臨床試験用品の市場を調査し、市場概要、市場影響因子および市場機会の分析、技術・特許動向、ケーススタディ、法規制環境、市場規模の推移・予測、各種区分・地域別の詳細分析、競合情勢、主要企業のプロファイルなどをまとめています。
目次
第1章 イントロダクション
第2章 調査手法
第3章 エグゼクティブサマリー
第4章 重要考察
第5章 市場概要
- 市場力学
- 臨床試験用品市場:影響分析
- 促進要因
- 抑制要因
- 機会
- 課題
- 顧客の事業に影響を与える動向/ディスラプション
- 価格分析
- 技術分析
- バリューチェーン分析
- エコシステム分析
- サプライチェーン分析
- 主な会議とイベント
- 規制分析
- ポーターのファイブフォース分析
- 主なステークホルダーと購入基準
第6章 臨床試験用品市場:サービス別
- 物流・流通
- 保管・保持
- 包装・ラベリング・ブラインディング
- 製造
- コンパレータ調達
- その他
第7章 臨床試験用品市場:フェーズ別
- フェーズⅢ
- フェーズII
- フェーズI
- フェーズIV
- BA/BE研究
第8章 臨床試験用品市場:タイプ別
- 低分子薬
- 生物学的製剤
- 医療機器
第9章 臨床試験用品市場:治療領域別
- 腫瘍
- 感染症
- 神経
- 代謝異常症
- 免疫
- 心臓病
- 遺伝性疾患・希少疾患
- その他
第10章 臨床試験用品市場:エンドユーザー別
- 製薬会社・バイオテクノロジー会社
- CRO
- 医療機器会社
第11章 臨床試験用品市場:地域別
- 北米
- 欧州
- アジア太平洋
- ラテンアメリカ
- 中東
- アフリカ
第12章 競合情勢
- 主要企業の採用戦略
- 収益シェア分析
- 市場シェア分析
- 企業評価マトリックス
- 企業のフットプリント
- 企業評価マトリックス:新興企業/中小企業
- 新興企業/中小企業の競合ベンチマーキング
- 競合シナリオと動向
第13章 企業プロファイル
- 主要企業
- THERMO FISHER SCIENTIFIC, INC.
- CATALENT, INC.
- EUROFINS SCIENTIFIC
- PIRAMAL PHARMA SOLUTIONS
- PRA HEALTH SCIENCES(A SUBSIDIARY OF ICON PLC)
- MARKEN(A SUBSIDIARY OF UPS)
- PAREXEL INTERNATIONAL(MA)CORPORATION
- BIOCAIR
- ALMAC GROUP
- SHARP SERVICES, LLC
- PCI PHARMA SERVICES
- NUVISAN
- LONZA GROUP
- その他の企業
- OCT GROUP LLC
- COREX LOGISTICS LIMITED
- ACNOS PHARMA GMBH
- CLINICAL SERVICES INTERNATIONAL
- KLIFO
- CLINIGEN LIMITED
- ANCILLARE LP
- N-SIDE
- ADALLEN PHARMA
- DURBIN
- RECIPHARM AB
- IPS PHARMA
第14章 付録
The global clinical trial supplies market size is projected to reach USD 6.3 billion by 2028 from USD 3.9 billion in 2023, at a CAGR of 9.9% during the forecast period. Factors such as Increasing number of clinical trials, Growing product pipeline, and Increasing decentralization of clinical trials are likely to have positive impact on the market growth.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2021-2028 |
| Base Year | 2022 |
| Forecast Period | 2023-2028 |
| Units Considered | Value (USD) billion |
| Segments | By Service, By Phase, By Type, By Therapeutic Area, By End User |
| Regions covered | North America, Europe, Asia Pacific, Latin America, Middle East and the Africa. |
"The logistics and distribution segment held the dominant share in the clinical trial supplies market"
Based on services, the global clinical trial supplies market is segmented into pathogen component, adjuvant emulsions, particulate adjuvants, saponin-based adjuvants and other adjuvants. The adjuvant emulsions segment held a dominant share in the market in 2022. Extensive use of adjuvant emulsions in COVID-19 & other infectious diseases and the rising prevalence of contagious diseases are likely to have positive impact on the growth of the market.
"Phase II segment accounted for the largest share of the phase segment in 2022."
Based on phase, the clinical trial supplies market is segmented into phase I, phase II, phase III, phase IV, BA/BE studies. In 2022, the phase II segment accounted for the largest share of the clinical trial supplies market. The segment held the dominant share in the market owing to various factors such as increasing the number of registration in clinical trials and the rising R&D investment by pharmaceutical and biotechnologies company for new cancer drug development.
"Asia Pacific region is likely to grow at a faster pace in the clinical trial supplies market."
The clinical trial supplies market region is segmented into North America, Europe, Asia Pacific, Latin America Middle East and the Africa. Asia Pacific region is anticipated to grow at a significant CAGR during the forecast period. The growth of the region is majorly driven by the increasing number of registered clinical trials. The increasing decentralization of clinical trials from China, India, and Japan due to presence of prominent players are engaged in providing services for clinical trial, supports market growth in the Asia Pacific.
The primary interviews conducted for this report can be categorized as follows:
- By Respondent: Supply Side- 80%, and Demand Side - 20%
- By Designation (Supply Side): Managers - 45%, CXOs & Directors - 30%, Executives- 25%
- By Region: North America -40%, Europe -25%, Asia-Pacific -20%, Latin America -10%, and Middle East and Africa -5%
List of Companies Profiled in the Report:
- Thermo Fisher Scientific Inc. (US)
- Catalent, Inc. (US)
- Eurofins (France)
- Piramal Pharma Solutions (India)
- PRA Health Sciences (Subsidiary of Icon Plc) (US)
- Marken (Subsidiary of UPS) (US)
- Parexel International (MA) Corporation (US)
- Biocair (UK)
- Almac Group (UK)
- Sharp Services, LLC (US)
- PCI Pharma Services (US)
- Nuvisan (Germany)
- Lonza Group (Switzerland)
- OCT Group LLC. (Latvia)
- Corex Logistics (Ireland)
- Acnos Pharma GMBH (Germany)
- Clinical Services International (UK)
- KLIFO A/S (Denmark)
- Clinigen Limited (UK)
- Ancillare LP (US)
- N-SIDE (Belgium)
- ADAllen Pharma (UK)
- Durbin ( Part of the Uniphar Group) (UK)
- Recipharm AB (Sweden)
- IPS Pharma (UK)
Research Coverage:
This report provides a detailed picture of the clinical trial supplies market. It aims at estimating the size and future growth potential of the market across different segments, such as service, phase, type, therapeutic area, end user and region. The report also includes an in-depth competitive analysis of the key market players, along with their company profiles, recent developments, and key market strategies.
Key Benefits of Buying the Report:
The report will help market leaders/new entrants by providing them with the closest approximations of the revenue numbers for the overall clinical trial supplies market and its subsegments. It will also help stakeholders better understand the competitive landscape and gain more insights to better position their business and make suitable go-to-market strategies. This report will enable stakeholders to understand the market's pulse and provide them with information on the key market drivers, restraints, opportunities, and challenges.
The report provides insights on the following pointers:
- Analysis of key drivers (Increasing R&D expenditure of pharmaceutical and biopharmaceutical companies, Increasing number of clinical trials, Growing decentralization of clinical trials, Rising prevalence of orphan and rare diseases, Increasing outsourcing activities in emerging Asian economies), restraints ( High cost of drug development), opportunities (Growth opportunities in emerging markets, Increased investments in development of biologics and biosimilars, Rising demand for specialized testing services) and challenges (High Cost of Clinical Trials) are influencing the growth of clinical trial supplies market.
- Service Development/Innovation: Detailed insights on newly launched services of the clinical trial supplies market.
- Market Development: Comprehensive information about lucrative markets - the report analyses the clinical trial supplies market across varied regions.
- Market Diversification: Exhaustive information about new services, untapped geographies, recent developments, and investments in the clinical trial supplies market.
Competitive Assessment: In-depth assessment of market shares, growth strategies, and service offerings of leading players include Thermo Fisher Scientific Inc. (US), Marken (US), Catalent, Inc. (US), Eurofins Scientific (France), and PRA Health Sciences (US), Parexel International (MA) Corporation (US), Biocair (UK), AlmacGroup (UK), Piramal Pharma Solutions (India), Sharp Services, LLC (US), PCI Pharma Services (US),
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.2.1 INCLUSIONS & EXCLUSIONS
- 1.3 MARKET SCOPE
- 1.3.1 MARKETS COVERED
- 1.3.2 YEARS CONSIDERED
- 1.4 CURRENCY CONSIDERED
- 1.5 RESEARCH LIMITATIONS
- 1.6 STAKEHOLDERS
- 1.7 SUMMARY OF CHANGES
- 1.8 RECESSION IMPACT
2 RESEARCH METHODOLOGY
- 2.1 RESEARCH APPROACH
- FIGURE 1 RESEARCH DESIGN
- 2.1.1 PRIMARY RESEARCH
- FIGURE 2 CLINICAL TRIAL SUPPLIES MARKET: PRIMARY RESPONDENTS
- 2.1.2 SECONDARY DATA
- 2.2 MARKET SIZE ESTIMATION
- FIGURE 3 CLINICAL TRIAL SUPPLIES MARKET SIZE ESTIMATION (SUPPLY-SIDE ANALYSIS), 2022
- FIGURE 4 MARKET SIZE ESTIMATION: APPROACH 1 (COMPANY REVENUE ANALYSIS-BASED ESTIMATION), 2022
- 2.2.1 INSIGHTS FROM PRIMARY
- FIGURE 5 MARKET SIZE VALIDATION FROM PRIMARY EXPERTS
- 2.2.2 SEGMENTAL ASSESSMENT (BY SERVICE, PHASE, TYPE, THERAPEUTIC AREA, END USER, AND REGION)
- FIGURE 6 MARKET SIZE ESTIMATION METHODOLOGY: TOP-DOWN APPROACH
- 2.3 GROWTH RATE ASSUMPTIONS
- FIGURE 7 CLINICAL TRIAL SUPPLIES MARKET (DEMAND SIDE): GROWTH ANALYSIS OF DEMAND-SIDE FACTORS
- FIGURE 8 CLINICAL TRIAL SUPPLIES MARKET: GROWTH ANALYSIS OF DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
- FIGURE 9 DATA TRIANGULATION METHODOLOGY
- 2.4 STUDY ASSUMPTIONS
- 2.5 RISK ANALYSIS
- 2.6 RECESSION IMPACT ANALYSIS
- TABLE 1 GLOBAL INFLATION RATE PROJECTIONS, 2021-2028 (% GROWTH)
- TABLE 2 US HEALTH EXPENDITURE, 2019-2022 (USD MILLION)
- TABLE 3 US HEALTH EXPENDITURE, 2023-2027 (USD MILLION)
3 EXECUTIVE SUMMARY
- FIGURE 10 CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2023 VS. 2028 (USD MILLION)
- FIGURE 11 CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2023 VS. 2028 (USD MILLION)
- FIGURE 12 CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2023 VS. 2028 (USD MILLION)
- FIGURE 13 CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2023 VS. 2028 (USD MILLION)
- FIGURE 14 CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2023 VS. 2028 (USD MILLION)
- FIGURE 15 GEOGRAPHICAL SNAPSHOT OF CLINICAL TRIAL SUPPLIES MARKET
4 PREMIUM INSIGHTS
- 4.1 CLINICAL TRIAL SUPPLIES MARKET OVERVIEW
- FIGURE 16 RISING PREVALENCE OF INFECTIOUS DISEASES TO DRIVE MARKET GROWTH
- 4.2 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE AND COUNTRY (2022)
- FIGURE 17 SMALL-MOLECULE DRUGS ACCOUNTED FOR LARGEST SHARE OF NORTH AMERICAN CLINICAL TRIAL SUPPLIES MARKET IN 2022
- 4.3 CLINICAL TRIAL SUPPLIES MARKET SHARE, BY SERVICE, 2023 VS. 2028
- FIGURE 18 LOGISTICS & DISTRIBUTION SERVICES TO CONTINUE TO DOMINATE MARKET IN 2028
- 4.4 CLINICAL TRIAL SUPPLIES MARKET SHARE, BY END USER, 2022
- FIGURE 19 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES ACCOUNTED FOR LARGEST MARKET SHARE IN 2022
- 4.5 CLINICAL TRIAL SUPPLIES MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
- FIGURE 20 ASIA PACIFIC COUNTRIES TO REGISTER HIGHEST GROWTH RATES FROM 2023 TO 2028
5 MARKET OVERVIEW
- 5.1 INTRODUCTION
- 5.2 MARKET DYNAMICS
- FIGURE 21 CLINICAL TRIAL SUPPLIES MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
- 5.3 CLINICAL TRIAL SUPPLIES MARKET: IMPACT ANALYSIS
- 5.3.1 DRIVERS
- 5.3.1.1 Increasing R&D investments by biopharma companies
- TABLE 4 R&D INVESTMENTS BY MAJOR PLAYERS (2022)
- 5.3.1.2 Growing number of clinical trials
- FIGURE 22 NUMBER OF REGISTERED CLINICAL TRIALS (2011-2022)
- FIGURE 23 ONGOING CLINICAL TRIALS, BY PHASE (% SHARE), 2022
- 5.3.1.3 Growing decentralization of clinical trials
- 5.3.1.4 Rising prevalence of orphan and rare diseases
- FIGURE 24 OVERVIEW OF TOP 20 COMPANIES FOCUSING ON RARE DISEASES (AS OF 2023)
- FIGURE 25 PERCENTAGE OF NEW ACTIVE SUBSTANCES LAUNCHED WITH ORPHAN DRUG DESIGNATION, 2012-2022
- 5.3.1.5 Increasing outsourcing activities in emerging Asian economies
- 5.3.2 RESTRAINTS
- 5.3.2.1 High cost of drug development
- 5.3.3 OPPORTUNITIES
- 5.3.3.1 Growth opportunities in emerging markets
- 5.3.3.2 Increased investments in development of biologics and biosimilars
- FIGURE 26 TOTAL NUMBER OF NEW DRUGS (NEW CHEMICAL ENTITIES AND BIOLOGICS) APPROVED BY US FDA (2015-2022)
- TABLE 5 LIST OF BIOLOGICS APPROVED BY US FDA, 2022
- FIGURE 27 BIOSIMILAR APPROVALS AND LAUNCHES (2015-2022)
- 5.3.3.3 Rising demand for specialized testing services
- 5.3.4 CHALLENGES
- 5.3.4.1 High cost of clinical trials
- 5.3.1 DRIVERS
- 5.4 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS' BUSINESSES
- FIGURE 28 REVENUE SHIFT AND NEW REVENUE POCKETS FOR CLINICAL TRIAL SERVICE PROVIDERS
- 5.5 PRICING ANALYSIS
- 5.5.1 INDICATIVE PRICING ANALYSIS OF OVERALL CLINICAL TRIALS, BY PHASE.
- TABLE 6 PHASE III ONCOLOGY CLINICAL TRIAL COST (US AND EUROPEAN COUNTRIES)
- TABLE 7 PHASE I ONCOLOGY CLINICAL TRIAL COST (SPAIN)
- 5.6 TECHNOLOGY ANALYSIS
- 5.6.1 INTERACTIVE RESPONSE TECHNOLOGY (IRT)
- 5.7 VALUE CHAIN ANALYSIS
- FIGURE 29 VALUE CHAIN ANALYSIS: RAW MATERIAL AND MANUFACTURING PHASES CONTRIBUTE MAXIMUM VALUE
- 5.8 ECOSYSTEM ANALYSIS
- 5.9 SUPPLY CHAIN ANALYSIS
- TABLE 8 CLINICAL TRIAL SUPPLIES MARKET: SUPPLY CHAIN ANALYSIS
- 5.10 KEY CONFERENCES & EVENTS IN 2023-2024
- TABLE 9 CLINICAL TRIAL SUPPLIES MARKET: DETAILED LIST OF CONFERENCES & EVENTS
- 5.11 REGULATORY ANALYSIS
- 5.11.1 REGULATORY LANDSCAPE FOR CLINICAL TRIAL SUPPLIES
- 5.11.1.1 North America
- 5.11.1.2 Europe
- 5.11.1.3 Asia Pacific
- 5.11.1.4 Rest of the World
- 5.11.2 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 10 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 11 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 12 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 13 LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 14 MIDDLE EAST & AFRICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 5.11.1 REGULATORY LANDSCAPE FOR CLINICAL TRIAL SUPPLIES
- 5.12 PORTER'S FIVE FORCES ANALYSIS
- TABLE 15 CLINICAL TRIAL SUPPLIES MARKET: PORTER'S FIVE FORCES ANALYSIS
- 5.12.1 THREAT OF NEW ENTRANTS
- 5.12.2 THREAT OF SUBSTITUTES
- 5.12.3 BARGAINING POWER OF SUPPLIERS
- 5.12.4 BARGAINING POWER OF BUYERS
- 5.12.5 DEGREE OF COMPETITION
- 5.13 KEY STAKEHOLDERS & BUYING CRITERIA
- 5.13.1 KEY STAKEHOLDERS IN BUYING PROCESS
- FIGURE 30 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS OF CLINICAL TRIAL SUPPLY SERVICES
- 5.13.2 BUYING CRITERIA FOR CLINICAL TRIAL SUPPLY SERVICES
- FIGURE 31 KEY BUYING CRITERIA FOR END USERS
6 CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE
- 6.1 INTRODUCTION
- TABLE 16 CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- 6.2 LOGISTICS & DISTRIBUTION
- 6.2.1 INCREASING COMPLEXITY AND EVOLVING TREATMENT PROTOCOLS TO DRIVE DEMAND FOR LOGISTICS & DISTRIBUTION SERVICES
- TABLE 17 CLINICAL TRIAL SUPPLIES MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 18 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 19 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 20 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 21 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR LOGISTICS & DISTRIBUTION SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- 6.3 STORAGE & RETENTION
- 6.3.1 GROWING DEMAND FOR DIRECT-TO-PATIENT AND DIRECT-TO-SITE SERVICES TO DRIVE GROWTH
- TABLE 22 CLINICAL TRIAL SUPPLIES MARKET FOR STORAGE & RETENTION SERVICES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 23 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR STORAGE & RETENTION SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 24 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR STORAGE & RETENTION SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 25 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR STORAGE & RETENTION SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 26 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR STORAGE & RETENTION SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- 6.4 PACKAGING, LABELING, AND BLINDING
- 6.4.1 RISING DEMAND FOR ADVANCED PACKAGING TO DRIVE SEGMENTAL GROWTH
- TABLE 27 CLINICAL TRIAL SUPPLIES MARKET FOR PACKAGING, LABELING, AND BLINDING SERVICES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 28 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PACKAGING, LABELING, AND BLINDING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 29 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR PACKAGING, LABELING, AND BLINDING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 30 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR PACKAGING, LABELING, AND BLINDING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 31 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR LABELING, AND BLINDING SERVICES, COUNTRY, 2021-2028 (USD MILLION)
- 6.5 MANUFACTURING
- 6.5.1 GROWING FOCUS OF COMPANIES ON INCREASING MANUFACTURING CAPACITIES TO DRIVE MARKET GROWTH
- TABLE 32 CLINICAL TRIAL SUPPLIES MARKET FOR MANUFACTURING SERVICES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 33 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR MANUFACTURING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 34 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR MANUFACTURING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 35 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR MANUFACTURING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 36 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR MANUFACTURING SERVICES, COUNTRY, 2021-2028 (USD MILLION)
- 6.6 COMPARATOR SOURCING
- 6.6.1 INTENSE MARKET COMPETITION AND RISING NUMBER OF CLINICAL TRIALS IN EMERGING COUNTRIES TO DRIVE GROWTH
- TABLE 37 CLINICAL TRIAL SUPPLIES MARKET FOR COMPARATOR SOURCING SERVICES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 38 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR COMPARATOR SOURCING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 39 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR COMPARATOR SOURCING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 40 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR COMPARATOR SOURCING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 41 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR COMPARATOR SOURCING SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- 6.7 OTHER SERVICES
- TABLE 42 CLINICAL TRIAL SUPPLIES MARKET FOR OTHER SERVICES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 43 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR OTHER SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 44 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR OTHER SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 45 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR OTHER SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 46 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR OTHER SERVICES, BY COUNTRY, 2021-2028 (USD MILLION)
7 CLINICAL TRIAL SUPPLIES MARKET, BY PHASE
- 7.1 INTRODUCTION
- TABLE 47 CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- 7.2 PHASE III
- 7.2.1 LARGE PATIENT POPULATION ASSOCIATED WITH PHASE III TRIALS TO DRIVE DEMAND
- TABLE 48 CLINICAL TRIAL SUPPLIES MARKET FOR PHASE III CLINICAL TRIALS, BY REGION, 2021-2028 (USD MILLION)
- TABLE 49 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE III CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 50 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE III CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 51 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE III CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 52 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE III CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- 7.3 PHASE II
- 7.3.1 ABILITY OF PHASE II CLINICAL TRIALS TO PROVIDE CLARITY ON EFFECTIVENESS AND SAFETY OF DRUGS TO DRIVE MARKET GROWTH
- TABLE 53 CLINICAL TRIAL SUPPLIES MARKET FOR PHASE II CLINICAL TRIALS, BY REGION, 2021-2028 (USD MILLION)
- TABLE 54 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE II CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 55 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE II CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 56 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE II CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 57 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE II CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- 7.4 PHASE I
- 7.4.1 IMPORTANCE OF QUALITY CLINICAL TRIAL SUPPLIES IN PHASE I CLINICAL TRIALS TO BOOST MARKET
- TABLE 58 CLINICAL TRIAL SUPPLIES MARKET FOR PHASE I CLINICAL TRIALS, BY REGION, 2021-2028 (USD MILLION)
- TABLE 59 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE I CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 60 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE I CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
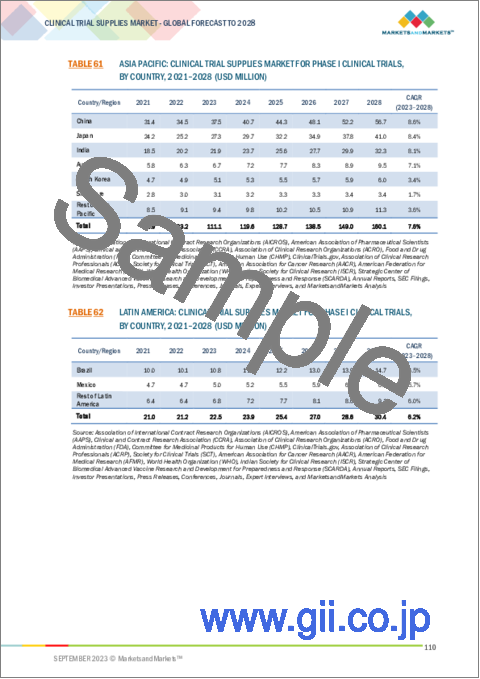
- TABLE 61 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE I CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 62 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE I CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- 7.5 PHASE IV
- 7.5.1 INCREASING NUMBER OF STUDIES UNDER PHASE IV TRIAL TO SUPPORT GROWTH
- TABLE 63 CLINICAL TRIAL SUPPLIES MARKET FOR PHASE IV CLINICAL TRIALS, BY REGION, 2021-2028 (USD MILLION)
- TABLE 64 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE IV CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 65 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE IV CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 66 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE IV CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 67 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHASE IV CLINICAL TRIALS, BY COUNTRY, 2021-2028 (USD MILLION)
- 7.6 BA/BE STUDIES
- 7.6.1 INCREASING PATENT EXPIRATION TO INCREASE DEMAND FOR BA/BE STUDIES
- TABLE 68 CLINICAL TRIAL SUPPLIES MARKET FOR BA/BE STUDIES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 69 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR BA/BE STUDIES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 70 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR BA/BE STUDIES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 71 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR BA/BE STUDIES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 72 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR BA/BE STUDIES, BY COUNTRY, 2021-2028 (USD MILLION)
8 CLINICAL TRIAL SUPPLIES MARKET, BY TYPE
- 8.1 INTRODUCTION
- TABLE 73 CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- 8.2 SMALL-MOLECULE DRUGS
- 8.2.1 LOW COST OF DEVELOPING SMALL-MOLECULE DRUGS TO DRIVE MARKET GROWTH
- TABLE 74 CLINICAL TRIAL SUPPLIES MARKET FOR SMALL-MOLECULE DRUGS, BY REGION, 2021-2028 (USD MILLION)
- TABLE 75 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR SMALL-MOLECULE DRUGS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 76 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR SMALL-MOLECULE DRUGS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 77 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR SMALL-MOLECULE DRUGS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 78 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR SMALL-MOLECULE DRUGS, BY COUNTRY, 2021-2028 (USD MILLION)
- 8.3 BIOLOGIC DRUGS
- 8.3.1 RELATIVELY LOW RATE OF ATTRITION OF BIOLOGIC DRUGS COMPARED TO SMALL-MOLECULE DRUGS TO BOOST GROWTH
- TABLE 79 CLINICAL TRIAL SUPPLIES MARKET FOR BIOLOGIC DRUGS, BY REGION, 2021-2028 (USD MILLION)
- TABLE 80 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR BIOLOGIC DRUGS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 81 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR BIOLOGIC DRUGS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 82 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR BIOLOGIC DRUGS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 83 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR BIOLOGIC DRUGS, BY COUNTRY, 2021-2028 (USD MILLION)
- 8.4 MEDICAL DEVICES
- 8.4.1 INCREASING NUMBER OF TRIALS FOR MEDICAL DEVICES TO SUPPORT GROWTH
- TABLE 84 CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 85 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 86 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 87 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 88 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICES, BY COUNTRY, 2021-2028 (USD MILLION)
9 CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA
- 9.1 INTRODUCTION
- TABLE 89 CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- 9.2 ONCOLOGY
- 9.2.1 GROWING STUDIES ON CANCER THERAPEUTICS TO DRIVE MARKET GROWTH
- FIGURE 32 US CANCER INCIDENCE IN MALES, BY TYPE (2022)
- FIGURE 33 US CANCER INCIDENCE IN FEMALES, BY TYPE (2022)
- FIGURE 34 GLOBAL ONCOLOGY-RELATED CLINICAL TRIALS, 2010-2021 (THOUSAND)
- TABLE 90 NEW FDA-APPROVED ONCOLOGY DRUGS (2023)
- TABLE 91 CLINICAL TRIAL SUPPLIES MARKET FOR ONCOLOGY, BY REGION, 2021-2028 (USD MILLION)
- TABLE 92 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR ONCOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 93 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR ONCOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 94 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR ONCOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 95 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR ONCOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- 9.3 INFECTIOUS DISEASES
- 9.3.1 INCREASING CASES OF INFECTIOUS DISEASES TO ACCELERATE EFFORTS TO DEVELOP NEW DRUGS
- TABLE 96 CLINICAL TRIAL SUPPLIES MARKET FOR INFECTIOUS DISEASES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 97 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR INFECTIOUS DISEASES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 98 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR INFECTIOUS DISEASES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 99 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR INFECTIOUS DISEASES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 100 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR INFECTIOUS DISEASES, BY COUNTRY, 2021-2028 (USD MILLION)
- 9.4 NEUROLOGY
- 9.4.1 GROWING INVESTMENTS BY PHARMACEUTICAL COMPANIES FOR NEUROLOGY RESEARCH TO DRIVE GROWTH
- TABLE 101 LIST OF PIPELINE DRUGS FOR NEUROLOGICAL DISORDERS (2020)
- TABLE 102 CLINICAL TRIAL SUPPLIES MARKET FOR NEUROLOGY, BY REGION, 2021-2028 (USD MILLION)
- TABLE 103 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR NEUROLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 104 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR NEUROLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 105 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR NEUROLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 106 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR NEUROLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- 9.5 METABOLIC DISORDERS
- 9.5.1 INCREASING PREVALENCE OF DIABETES AND OTHER METABOLIC DISORDERS TO DRIVE MARKET GROWTH
- FIGURE 35 ESTIMATED NUMBER OF INDIVIDUALS WITH DIABETES, 2000-2045 (MILLION)
- TABLE 107 CLINICAL TRIAL SUPPLIES MARKET FOR METABOLIC DISORDERS, BY REGION, 2021-2028 (USD MILLION)
- TABLE 108 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR METABOLIC DISORDERS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 109 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR METABOLIC DISORDERS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 110 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR METABOLIC DISORDERS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 111 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR METABOLIC DISORDERS, BY COUNTRY, 2021-2028 (USD MILLION)
- 9.6 IMMUNOLOGY
- 9.6.1 CONTINUOUS GROWTH IN CLINICAL STUDIES FOR IMMUNE SYSTEM DISEASES TO FUEL DEMAND FOR CLINICAL TRIAL SUPPLIES
- TABLE 112 LIST OF PIPELINE DRUGS FOR IMMUNOLOGICAL DISORDERS (2022)
- TABLE 113 CLINICAL TRIAL SUPPLIES MARKET FOR IMMUNOLOGY, BY REGION, 2021-2028 (USD MILLION)
- TABLE 114 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR IMMUNOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 115 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR IMMUNOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 116 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR IMMUNOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 117 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR IMMUNOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- 9.7 CARDIOLOGY
- 9.7.1 GROWING INVESTMENTS BY PHARMACEUTICAL COMPANIES FOR CVD RESEARCH TO DRIVE GROWTH
- TABLE 118 LIST OF PIPELINE DRUGS FOR CARDIOVASCULAR DISEASES (2023)
- TABLE 119 CLINICAL TRIAL SUPPLIES MARKET FOR CARDIOLOGY, BY REGION, 2021-2028 (USD MILLION)
- TABLE 120 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR CARDIOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 121 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR CARDIOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 122 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR CARDIOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 123 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR CARDIOLOGY, BY COUNTRY, 2021-2028 (USD MILLION)
- 9.8 GENETIC & RARE DISEASES
- 9.8.1 INCREASING R&D FOR DEVELOPMENT OF EFFECTIVE DRUGS TO DRIVE DEMAND
- TABLE 124 CLINICAL TRIAL SUPPLIES MARKET FOR GENETIC & RARE DISEASES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 125 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR GENETIC & RARE DISEASES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 126 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR GENETIC & RARE DISEASES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 127 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR GENETIC & RARE DISEASES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 128 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR GENETIC & RARE DISEASES, BY COUNTRY, 2021-2028 (USD MILLION)
- 9.9 OTHER THERAPEUTIC AREAS
- TABLE 129 LIST OF PIPELINE DRUGS FOR SKIN DISEASES (2022)
- TABLE 130 CLINICAL TRIAL SUPPLIES MARKET FOR OTHER THERAPEUTIC AREAS, BY REGION, 2021-2028 (USD MILLION)
- TABLE 131 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR OTHER THERAPEUTIC AREAS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 132 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR OTHER THERAPEUTIC AREAS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 133 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR OTHER THERAPEUTIC AREAS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 134 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR OTHER THERAPEUTIC AREAS, BY COUNTRY, 2021-2028 (USD MILLION)
10 CLINICAL TRIAL SUPPLIES MARKET, BY END USER
- 10.1 INTRODUCTION
- TABLE 135 CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 10.2 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES
- 10.2.1 INCREASING OUTSOURCING OF CLINICAL TRIAL SUPPLIES BY PHARMA & BIOTECH COMPANIES TO BOOST MARKET
- TABLE 136 CLINICAL TRIAL SUPPLIES MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 137 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 138 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 139 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 140 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES, BY COUNTRY, 2021-2028 (USD MILLION)
- 10.3 CONTRACT RESEARCH ORGANIZATIONS
- 10.3.1 INCREASING OUTSOURCING OF CLINICAL TRIALS TO DRIVE ADOPTION OF CLINICAL TRIAL SUPPLIES AMONG CROS
- TABLE 141 CLINICAL TRIAL SUPPLIES MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY REGION, 2021-2028 (USD MILLION)
- TABLE 142 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 143 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 144 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 145 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR CONTRACT RESEARCH ORGANIZATIONS, BY COUNTRY, 2021-2028 (USD MILLION)
- 10.4 MEDICAL DEVICE COMPANIES
- 10.4.1 INCREASING NUMBER OF TRIALS OF MEDICAL DEVICES TO DRIVE MARKET
- TABLE 146 CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICE COMPANIES, BY REGION, 2021-2028 (USD MILLION)
- TABLE 147 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICE COMPANIES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 148 EUROPE: CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICE COMPANIES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 149 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICE COMPANIES, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 150 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET FOR MEDICAL DEVICE COMPANIES, BY COUNTRY, 2021-2028 (USD MILLION)
11 CLINICAL TRIAL SUPPLIES MARKET, BY REGION
- 11.1 INTRODUCTION
- TABLE 151 CLINICAL TRIAL SUPPLIES MARKET, BY REGION, 2021-2028 (USD MILLION)
- 11.2 NORTH AMERICA
- FIGURE 36 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET SNAPSHOT
- TABLE 152 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 153 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 154 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 155 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 156 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 157 NORTH AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.2.1 US
- 11.2.1.1 Increasing R&D investments to drive market growth in US
- TABLE 158 US: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 159 US: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 160 US: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 161 US: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 162 US: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.2.2 CANADA
- 11.2.2.1 Growing preference of pharmaceutical companies to conduct trials in Canada to favor market growth
- TABLE 163 CANADA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 164 CANADA: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 165 CANADA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 166 CANADA: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 167 CANADA: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.2.3 NORTH AMERICA: RECESSION IMPACT
- 11.3 EUROPE
- TABLE 168 EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 169 EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 170 EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 171 EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 172 EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 173 EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.3.1 GERMANY
- 11.3.1.1 Growing number of sponsored trials to boost demand for clinical trial supplies in Germany
- TABLE 174 GERMANY: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 175 GERMANY: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 176 GERMANY: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 177 GERMANY: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 178 GERMANY: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.3.2 UK
- 11.3.2.1 Government policies leading to costlier R&D and supply chain restrictions post Brexit to limit market growth
- TABLE 179 UK: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 180 UK: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 181 UK: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 182 UK: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 183 UK: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.3.3 FRANCE
- 11.3.3.1 Presence of leading pharmaceutical companies to support market growth in France
- TABLE 184 FRANCE: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 185 FRANCE: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 186 FRANCE: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 187 FRANCE: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 188 FRANCE: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.3.4 ITALY
- 11.3.4.1 Increasing number of drug approvals to drive market for clinical trial supplies in Italy
- TABLE 189 ITALY: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 190 ITALY: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 191 ITALY: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 192 ITALY: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 193 ITALY: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.3.5 SPAIN
- 11.3.5.1 Increasing biologics production to support market growth in Spain
- TABLE 194 SPAIN: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 195 SPAIN: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 196 SPAIN: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 197 SPAIN: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 198 SPAIN: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.3.6 SWITZERLAND
- 11.3.6.1 Growing R&D efforts to provide growth opportunities for clinical trial supplies market in Switzerland
- TABLE 199 SWITZERLAND: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 200 SWITZERLAND: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 201 SWITZERLAND: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 202 SWITZERLAND: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 203 SWITZERLAND: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.3.7 NETHERLANDS
- 11.3.7.1 Increasing pharmaceutical production to boost clinical trial supplies market in Netherlands
- TABLE 204 NETHERLANDS: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 205 NETHERLANDS: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 206 NETHERLANDS: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 207 NETHERLANDS: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 208 NETHERLANDS: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.3.8 REST OF EUROPE
- TABLE 209 REST OF EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 210 REST OF EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 211 REST OF EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 212 REST OF EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 213 REST OF EUROPE: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.3.9 EUROPE: RECESSION IMPACT
- 11.4 ASIA PACIFIC
- FIGURE 37 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET SNAPSHOT
- TABLE 214 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY COUNTRY, 2021-2028 (USD MILLION)
- TABLE 215 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 216 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 217 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 218 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 219 ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.4.1 CHINA
- 11.4.1.1 Favorable government regulations to boost market growth in China
- TABLE 220 CHINA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 221 CHINA: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 222 CHINA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 223 CHINA: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 224 CHINA: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.4.2 JAPAN
- 11.4.2.1 Government initiatives for drug innovation to support market growth
- TABLE 225 JAPAN: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 226 JAPAN: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 227 JAPAN: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 228 JAPAN: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 229 JAPAN: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.4.3 INDIA
- 11.4.3.1 Low manufacturing costs and skilled workforce to attract outsourcing and investments to India
- TABLE 230 INDIA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 231 INDIA: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 232 INDIA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 233 INDIA: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 234 INDIA: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.4.4 AUSTRALIA
- 11.4.4.1 Presence of many research institutes conducting clinical trials to boost market growth
- TABLE 235 AUSTRALIA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 236 AUSTRALIA: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 237 AUSTRALIA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 238 AUSTRALIA: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 239 AUSTRALIA: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.4.5 SOUTH KOREA
- 11.4.5.1 Government investments to contribute to market growth in South Korea
- TABLE 240 SOUTH KOREA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 241 SOUTH KOREA: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 242 SOUTH KOREA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 243 SOUTH KOREA: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 244 SOUTH KOREA: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.4.6 SINGAPORE
- 11.4.6.1 Presence of world-class infrastructure to drive market growth in Singapore
- TABLE 245 SINGAPORE: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 246 SINGAPORE: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 247 SINGAPORE: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 248 SINGAPORE: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 249 SINGAPORE: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.4.7 REST OF ASIA PACIFIC
- TABLE 250 REST OF ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 251 REST OF ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 252 REST OF ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 253 REST OF ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 254 REST OF ASIA PACIFIC: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.4.8 ASIA PACIFIC: RECESSION IMPACT
- 11.5 LATIN AMERICA
- TABLE 255 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 256 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY REGION, 2021-2028 (USD MILLION)
- TABLE 257 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 258 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 259 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 260 LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.5.1 BRAZIL
- 11.5.1.1 Growth in pharmaceutical R&D to support market growth
- TABLE 261 BRAZIL: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 262 BRAZIL: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 263 BRAZIL: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 264 BRAZIL: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 265 BRAZIL: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.5.2 MEXICO
- 11.5.2.1 Growing number of Phase III clinical trials to drive market growth
- TABLE 266 MEXICO: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 267 MEXICO: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 268 MEXICO: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 269 MEXICO: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 270 MEXICO: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.5.3 REST OF LATIN AMERICA
- TABLE 271 REST OF LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 272 REST OF LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 273 REST OF LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 274 REST OF LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 275 REST OF LATIN AMERICA: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.5.4 LATIN AMERICA: RECESSION IMPACT
- 11.6 MIDDLE EAST
- 11.6.1 GROWING PRESENCE OF PHARMACEUTICAL COMPANIES TO DRIVE MARKET GROWTH
- TABLE 276 MIDDLE EAST: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 277 MIDDLE EAST: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 278 MIDDLE EAST: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 279 MIDDLE EAST: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 280 MIDDLE EAST: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.6.2 MIDDLE EAST: RECESSION IMPACT
- 11.7 AFRICA
- 11.7.1 GROWING DEMAND FOR OUTSOURCING SERVICES TO DRIVE MARKET GROWTH
- TABLE 281 AFRICA: CLINICAL TRIAL SUPPLIES MARKET, BY SERVICE, 2021-2028 (USD MILLION)
- TABLE 282 AFRICA: CLINICAL TRIAL SUPPLIES MARKET, BY PHASE, 2021-2028 (USD MILLION)
- TABLE 283 AFRICA: CLINICAL TRIAL SUPPLIES MARKET, BY TYPE, 2021-2028 (USD MILLION)
- TABLE 284 AFRICA: CLINICAL TRIAL SUPPLIES MARKET, BY THERAPEUTIC AREA, 2021-2028 (USD MILLION)
- TABLE 285 AFRICA: CLINICAL TRIAL SUPPLIES MARKET, BY END USER, 2021-2028 (USD MILLION)
- 11.7.2 AFRICA: RECESSION IMPACT
12 COMPETITIVE LANDSCAPE
- 12.1 INTRODUCTION
- 12.2 STRATEGIES ADOPTED BY KEY PLAYERS
- FIGURE 38 CLINICAL TRIAL SUPPLIES MARKET: STRATEGIES ADOPTED BY KEY PLAYERS
- 12.3 REVENUE SHARE ANALYSIS
- FIGURE 39 REVENUE ANALYSIS OF KEY PLAYERS (2020-2022)
- 12.4 MARKET SHARE ANALYSIS
- FIGURE 40 MARKET SHARE ANALYSIS OF KEY PLAYERS (2022)
- TABLE 286 CLINICAL TRIAL SUPPLIES MARKET: DEGREE OF COMPETITION
- 12.5 COMPANY EVALUATION MATRIX
- FIGURE 41 CLINICAL TRIAL SUPPLIES MARKET: COMPANY EVALUATION MATRIX FOR KEY PLAYERS, 2022
- 12.5.1 STARS
- 12.5.2 EMERGING LEADERS
- 12.5.3 PERVASIVE PLAYERS
- 12.5.4 PARTICIPANTS
- 12.6 COMPANY FOOTPRINT
- 12.6.1 SERVICE FOOTPRINT OF TOP 25 COMPANIES
- TABLE 287 CLINICAL TRIAL SUPPLIES MARKET: COMPANY SERVICE FOOTPRINT ANALYSIS OF KEY PLAYERS
- 12.6.2 REGIONAL FOOTPRINT OF TOP 25 COMPANIES
- TABLE 288 CLINICAL TRIAL SUPPLIES MARKET: REGIONAL FOOTPRINT ANALYSIS OF KEY PLAYERS
- 12.7 COMPANY EVALUATION MATRIX: START-UPS/SMES
- FIGURE 42 CLINICAL TRIAL SUPPLIES MARKET: COMPANY EVALUATION MATRIX FOR START-UPS/SMES, 2022
- 12.7.1 PROGRESSIVE COMPANIES
- 12.7.2 STARTING BLOCKS
- 12.7.3 RESPONSIVE COMPANIES
- 12.7.4 DYNAMIC COMPANIES
- 12.8 COMPETITIVE BENCHMARKING OF START-UP/SME PLAYERS
- TABLE 289 CLINICAL TIAL SUPPLIES MARKET: DETAILED LIST OF KEY START-UP/SME PLAYERS
- TABLE 290 CLINICAL TRIAL SUPPLIES MARKET: COMPETITIVE BENCHMARKING OF START-UPS/SME PLAYERS
- 12.9 COMPETITIVE SCENARIO AND TRENDS
- 12.9.1 SERVICES LAUNCHES & APPROVALS
- TABLE 291 CLINICAL TRIAL SUPPLIES MARKET: SERVICE LAUNCHES, JANUARY 2021-AUGUST 2023
- 12.9.2 DEALS
- TABLE 292 CLINICAL TRIAL SUPPLIES MARKET: DEALS, JANUARY 2021-JULY 2023
- 12.9.3 OTHER DEVELOPMENTS
- TABLE 293 CLINICAL TRIAL SUPPLIES MARKET: OTHER DEVELOPMENTS, JANUARY 2021-JULY 2023
13 COMPANY PROFILES
- (Business overview, Services offered, Recent Developments, MNM view)**
- 13.1 KEY MARKET PLAYERS
- 13.1.1 THERMO FISHER SCIENTIFIC, INC.
- TABLE 294 THERMO FISHER SCIENTIFIC: BUSINESS OVERVIEW
- FIGURE 43 THERMO FISHER SCIENTIFIC, INC.: COMPANY SNAPSHOT (2022)
- 13.1.2 CATALENT, INC.
- TABLE 295 CATALENT, INC.: BUSINESS OVERVIEW
- FIGURE 44 CATALENT, INC.: COMPANY SNAPSHOT (2022)
- 13.1.3 EUROFINS SCIENTIFIC
- TABLE 296 EUROFINS SCIENTIFIC: BUSINESS OVERVIEW
- FIGURE 45 EUROFINS SCIENTIFIC: COMPANY SNAPSHOT (2022)
- 13.1.4 PIRAMAL PHARMA SOLUTIONS
- TABLE 297 PIRAMAL PHARMA SOLUTIONS: BUSINESS OVERVIEW
- FIGURE 46 PIRAMAL PHARMA SOLUTIONS: COMPANY SNAPSHOT (2022)
- 13.1.5 PRA HEALTH SCIENCES (A SUBSIDIARY OF ICON PLC)
- TABLE 298 PRA HEALTH SCIENCES: BUSINESS OVERVIEW
- FIGURE 47 ICON PLC: COMPANY SNAPSHOT (2022)
- 13.1.6 MARKEN (A SUBSIDIARY OF UPS)
- TABLE 299 UPS: BUSINESS OVERVIEW
- FIGURE 48 UPS: COMPANY SNAPSHOT (2022)
- 13.1.7 PAREXEL INTERNATIONAL (MA) CORPORATION
- TABLE 300 PAREXEL INTERNATIONAL (MA) CORPORATION: BUSINESS OVERVIEW
- 13.1.8 BIOCAIR
- TABLE 301 BIOCAIR: BUSINESS OVERVIEW
- 13.1.9 ALMAC GROUP
- TABLE 302 ALMAC GROUP: BUSINESS OVERVIEW
- 13.1.10 SHARP SERVICES, LLC
- TABLE 303 SHARP SERVICES, LLC: BUSINESS OVERVIEW
- 13.1.11 PCI PHARMA SERVICES
- TABLE 304 PCI PHARMA SERVICES: BUSINESS OVERVIEW
- 13.1.12 NUVISAN
- TABLE 305 NUVISAN: BUSINESS OVERVIEW
- 13.1.13 LONZA GROUP
- TABLE 306 LONZA GROUP: BUSINESS OVERVIEW
- FIGURE 49 LONZA GROUP: COMPANY SNAPSHOT (2022)
- 13.2 OTHER PLAYERS
- 13.2.1 OCT GROUP LLC
- 13.2.2 COREX LOGISTICS LIMITED
- 13.2.3 ACNOS PHARMA GMBH
- 13.2.4 CLINICAL SERVICES INTERNATIONAL
- 13.2.5 KLIFO
- 13.2.6 CLINIGEN LIMITED
- 13.2.7 ANCILLARE LP
- 13.2.8 N-SIDE
- 13.2.9 ADALLEN PHARMA
- 13.2.10 DURBIN
- 13.2.11 RECIPHARM AB
- 13.2.12 IPS PHARMA
- *Details on Business overview, Services offered, Recent Developments, MNM view might not be captured in case of unlisted companies.
14 APPENDIX
- 14.1 DISCUSSION GUIDE
- 14.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 14.3 CUSTOMIZATION OPTIONS
- 14.4 RELATED REPORTS
- 14.5 AUTHOR DETAILS