|
|
市場調査レポート
商品コード
1261342
足・足首用デバイスの世界市場規模、シェア、産業動向分析レポート用途別、製品別、地域別展望と予測、2022年~2028年Global Foot and Ankle Devices Market Size, Share & Industry Trends Analysis Report By Application, By Product, By Regional Outlook and Forecast, 2022 - 2028 |
||||||
| 足・足首用デバイスの世界市場規模、シェア、産業動向分析レポート用途別、製品別、地域別展望と予測、2022年~2028年 |
|
出版日: 2023年03月31日
発行: KBV Research
ページ情報: 英文 224 Pages
納期: 即納可能
|
- 全表示
- 概要
- 図表
- 目次
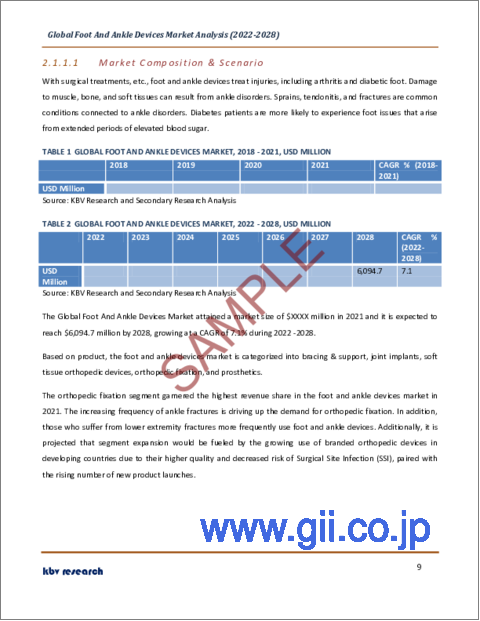
足・足首用デバイス市場規模は、2028年には61億米ドルに達し、予測期間中にCAGR7.1%の市場成長率で上昇すると予測されています。
関節リウマチ(RA)は、全世界でおよそ1%の人が罹患しており、女性は男性の2倍以上の頻度で罹患していると言われています。世界保健機関(WHO)によると、2,300万人以上の人々がRAに罹患していると考えられています。しかし、現在、RA患者さんが受けているヘルスケアは、今後予想される経済的負担に対応できるものではありません。専門家によると、2030年までにRA患者の数は倍増する可能性があります。これは、ベビーブーム世代の年齢層の移行、専門家によるRAへの理解の深まり、70歳以上のRA発症率がやや高くなることなどに起因すると考えられています。
さらに、世界中で2億人以上の人々が骨粗鬆症であると考えられています。国際骨粗鬆症財団の最新のデータでは、50歳以上の女性の3人に1人、男性の5人に1人が生涯のうちに骨粗鬆症性骨折を起こすとされています。また、骨粗鬆症は平均寿命を縮めるだけでなく、障害調整後の寿命も延ばし、そのケアを担う各国の医療保険制度に多大な財政負担を強いています。そのため、足部・足首用デバイスの市場は急速に拡大すると予想されています。
COVID-19の影響度分析
パンデミックの間、足首の外傷患者は大幅に減少しました。その結果、病院がCOVID-19の流行に目を向けるようになり、足部・足首用機器のニーズが減少しました。日常的な患者さんに集中できない結果、整形外科の専門家は苦しんでいました。COVID-19患者の治療には、医療従事者の再配置が必要でした。病院の従業員や運営インフラがないため、市場の拡大が遅れました。サプライチェーンが制約され、これらの機器の流通が妨げられ、世界中で治療が減少したため、COVID-19パンデミックは足と足首の機器市場に深刻な影響を与えました。
市場成長要因
世界的に増加する足と足首の問題
関節リウマチ、変形性関節症、外傷後関節症は、一般的に足と足首に影響を及ぼす3つの主要な関節炎です。足関節装具(AFO)のような装具を使用することで、運動能力を向上させることができます。また、装具や特殊な構造の装具を使用することで、痛みを軽減し、足への負担を軽減することができます。一般的な製品に比べ、有効性の向上、低コスト、患者の快適性、使用の利便性などの利点があるため、これらの機器の需要が増加し、足と足首の機器市場に利益をもたらすと予想されます。
テクノロジーと戦略的イニシアチブの導入の増加
また、患者数、手術の専門性、中国などの国も重要な指標を示しています。3Dプリント整形外科デバイスの分野は、コバルトクロム合金、ステンレス鋼、α-βチタン合金(Ti-6Al-4V)製の個別補綴物や金属製インプラントを含むことが特徴となっています。そのため、市場参入企業による戦略的な取り組みや、機器の製造における先進技術の受け入れが進んでいることが、市場の拡大を後押ししています。
市場抑制要因
フット&アンクルデバイスの重篤な副作用について
装具を長期間使用すると、靭帯、関節、周囲の筋肉が萎縮し、調子が悪くなることがあります。特に、患部を補強するための日常的な活動をさぼると、その傾向は顕著になります。関節の機能レベルが低下する、装具を装着していないときの関節の痛みや不快感が増す、日常生活で装具に頼ることが多くなる、などは装具の装着期間が長くなりすぎることの警告です。これらは、フット&アンクルデバイス市場全体の拡大にとって障害となる可能性があります。
製品の展望
製品に基づき、足部・足首デバイス市場は、ブレース&サポート、関節インプラント、軟組織整形外科デバイス、整形外科固定、補綴に分類されます。2021年のフット&アンクルデバイス市場では、ブレース&サポート部門がかなりの成長率を確保しました。このセグメントの上昇は、これらのデバイスの利点に起因しています。装具やサポーターなど、足や足首の周りに装着するデバイスは、サポートを提供することで関節を安定させるのに役立ちます。捻挫や骨折、腱鞘炎などの怪我をした場合、これらの器具を使用して治療することが多くあります。また、足や足首の問題を起こす危険性がある場合、装具を使用することで怪我を回避することができます。
アプリケーションの展望
用途別では、足部・足首デバイス市場は、ハンマートゥ、外傷、変形性関節症、関節リウマチ、神経障害、バニオン、骨粗鬆症に分けられます。骨粗鬆症セグメントは、2021年の足と足首のデバイス市場で顕著な成長率を獲得しました。このセグメントの拡大は、ほとんどの骨粗鬆症のケースでサポートデバイスの需要が高まっていることに起因しています。骨粗鬆症は足首の骨折を誘発する可能性があり、これは力学的に十分な仮定です。広範な疫学調査によると、BMIが高い男女は脆弱性骨折のリスクが低く、BMDも高いです。
地域別展望
地域別に見ると、足部・足首デバイス市場は、北米、欧州、アジア太平洋、LAMEAで分析されています。北米地域は、2021年に足と足首のデバイス市場で最も高い収益シェアを獲得しました。かなりの高齢者人口プールの普及と、スポーツや身体活動関連の怪我の頻度の高さが、この地域の大きな割合を占めている主な原因です。America's Health Rankings Senior Reportによると、米国では人口のおよそ16.9%にあたる約5,560万人の成人が65歳以上です。
市場参入企業がとる主な戦略は、製品の立ち上げです。カーディナルマトリックスに記載された分析によると、デピューシンセ(ジョンソン・エンド・ジョンソン)は、足と足首のデバイス市場における主要な前身です。Enovis Corporation、Stryker Corporation、Paragon 28, Inc.などの企業は、足と足首のデバイス市場における主要な革新的企業の一部です。
目次
第1章 市場範囲と調査手法
- 市場の定義
- 目的
- 市場範囲
- セグメンテーション
- 世界の足と足首のデバイス市場:用途別
- 世界の足と足首のデバイス市場:製品別
- 世界の足と足首のデバイス市場:地域別
- 調査手法
第2章 市場概要
- イントロダクション
- 概要
- 市場構成とシナリオ
- 概要
- 市場に影響を与える主な要因
- 市場促進要因
- 市場抑制要因
第3章 競合分析- 世界
- KBVカーディナルマトリックス
- 最近の業界全体の戦略的展開
- パートナーシップ、コラボレーション、および契約
- 製品の発売と製品の拡大
- 買収と合併
- 承認と試験
- 主要成功戦略
- 主要なリーディングストラテジー
- 主要な戦略的動き
第4章 世界の足と足首のデバイス市場:用途別
- 世界の外傷市場:地域別
- 世界の変形性関節症市場:地域別
- 世界の関節リウマチ市場:地域別
- 世界ハンマートー市場:地域別
- 世界の骨粗鬆症市場:地域別
- 世界の神経疾患市場:地域別
- 世界外反母趾市場:地域別
第5章 世界の足と足首のデバイス市場:製品別
- 世界の整形外科固定市場:地域別
- 世界の関節インプラント市場:地域別
- 世界ブレーシング&サポート市場:地域別
- 世界の補綴市場:地域別
- 世界の軟組織整形外科用デバイス市場:地域別
第6章 世界の足と足首のデバイス市場:地域別
- 北米
- 北米の市場:国別
- 米国
- カナダ
- メキシコ
- その他北米地域
- 北米の市場:国別
- 欧州
- 欧州の市場:国別
- ドイツ
- 英国
- フランス
- ロシア
- スペイン
- イタリア
- その他欧州地域
- 欧州の市場:国別
- アジア太平洋
- アジア太平洋の市場:国別
- 中国
- 日本
- インド
- 韓国
- シンガポール
- マレーシア
- その他アジア太平洋地域
- アジア太平洋の市場:国別
- ラテンアメリカ・中東・アフリカ
- ラテンアメリカ・中東・アフリカの市場:国別
- ブラジル
- アルゼンチン
- アラブ首長国連邦
- サウジアラビア
- 南アフリカ
- ナイジェリア
- その他ラテンアメリカ・中東・アフリカ地域
- ラテンアメリカ・中東・アフリカの市場:国別
第7章 企業プロファイル
- Stryker Corporation
- DePuy Synthes(Johnson & Johnson)
- Zimmer Biomet Holdings, Inc
- Smith & Nephew PLC
- Ossur Hf
- Enovis Corporation
- Acumed LLC(Colson Medical, Inc)
- Arthrex, Inc
- Paragon 28, Inc
- Orthofix Medical, Inc
LIST OF TABLES
- TABLE 1 Global Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 2 Global Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 3 Partnerships, Collaborations and Agreements- Foot and Ankle Devices Market
- TABLE 4 Product Launches And Product Expansions- Foot and Ankle Devices Market
- TABLE 5 Acquisition and Mergers- Foot and Ankle Devices Market
- TABLE 6 Approvals & Trials - Foot and Ankle Devices Market
- TABLE 7 Global Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 8 Global Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 9 Global Trauma Market by Region, 2018 - 2021, USD Million
- TABLE 10 Global Trauma Market by Region, 2022 - 2028, USD Million
- TABLE 11 Global Osteoarthritis Market by Region, 2018 - 2021, USD Million
- TABLE 12 Global Osteoarthritis Market by Region, 2022 - 2028, USD Million
- TABLE 13 Global Rheumatoid Arthritis Market by Region, 2018 - 2021, USD Million
- TABLE 14 Global Rheumatoid Arthritis Market by Region, 2022 - 2028, USD Million
- TABLE 15 Global Hammertoe Market by Region, 2018 - 2021, USD Million
- TABLE 16 Global Hammertoe Market by Region, 2022 - 2028, USD Million
- TABLE 17 Global Osteoporosis Market by Region, 2018 - 2021, USD Million
- TABLE 18 Global Osteoporosis Market by Region, 2022 - 2028, USD Million
- TABLE 19 Global Neurological Disorders Market by Region, 2018 - 2021, USD Million
- TABLE 20 Global Neurological Disorders Market by Region, 2022 - 2028, USD Million
- TABLE 21 Global Bunions Market by Region, 2018 - 2021, USD Million
- TABLE 22 Global Bunions Market by Region, 2022 - 2028, USD Million
- TABLE 23 Global Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 24 Global Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 25 Global Orthopedic Fixation Market by Region, 2018 - 2021, USD Million
- TABLE 26 Global Orthopedic Fixation Market by Region, 2022 - 2028, USD Million
- TABLE 27 Global Joint Implants Market by Region, 2018 - 2021, USD Million
- TABLE 28 Global Joint Implants Market by Region, 2022 - 2028, USD Million
- TABLE 29 Global Bracing & Support Market by Region, 2018 - 2021, USD Million
- TABLE 30 Global Bracing & Support Market by Region, 2022 - 2028, USD Million
- TABLE 31 Global Prosthetics Market by Region, 2018 - 2021, USD Million
- TABLE 32 Global Prosthetics Market by Region, 2022 - 2028, USD Million
- TABLE 33 Global Soft Tissue Orthopedic Devices Market by Region, 2018 - 2021, USD Million
- TABLE 34 Global Soft Tissue Orthopedic Devices Market by Region, 2022 - 2028, USD Million
- TABLE 35 Global Foot And Ankle Devices Market by Region, 2018 - 2021, USD Million
- TABLE 36 Global Foot And Ankle Devices Market by Region, 2022 - 2028, USD Million
- TABLE 37 North America Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 38 North America Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 39 North America Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 40 North America Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 41 North America Trauma Market by Country, 2018 - 2021, USD Million
- TABLE 42 North America Trauma Market by Country, 2022 - 2028, USD Million
- TABLE 43 North America Osteoarthritis Market by Country, 2018 - 2021, USD Million
- TABLE 44 North America Osteoarthritis Market by Country, 2022 - 2028, USD Million
- TABLE 45 North America Rheumatoid Arthritis Market by Country, 2018 - 2021, USD Million
- TABLE 46 North America Rheumatoid Arthritis Market by Country, 2022 - 2028, USD Million
- TABLE 47 North America Hammertoe Market by Country, 2018 - 2021, USD Million
- TABLE 48 North America Hammertoe Market by Country, 2022 - 2028, USD Million
- TABLE 49 North America Osteoporosis Market by Country, 2018 - 2021, USD Million
- TABLE 50 North America Osteoporosis Market by Country, 2022 - 2028, USD Million
- TABLE 51 North America Neurological Disorders Market by Country, 2018 - 2021, USD Million
- TABLE 52 North America Neurological Disorders Market by Country, 2022 - 2028, USD Million
- TABLE 53 North America Bunions Market by Country, 2018 - 2021, USD Million
- TABLE 54 North America Bunions Market by Country, 2022 - 2028, USD Million
- TABLE 55 North America Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 56 North America Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 57 North America Orthopedic Fixation Market by Country, 2018 - 2021, USD Million
- TABLE 58 North America Orthopedic Fixation Market by Country, 2022 - 2028, USD Million
- TABLE 59 North America Joint Implants Market by Country, 2018 - 2021, USD Million
- TABLE 60 North America Joint Implants Market by Country, 2022 - 2028, USD Million
- TABLE 61 North America Bracing & Support Market by Country, 2018 - 2021, USD Million
- TABLE 62 North America Bracing & Support Market by Country, 2022 - 2028, USD Million
- TABLE 63 North America Prosthetics Market by Country, 2018 - 2021, USD Million
- TABLE 64 North America Prosthetics Market by Country, 2022 - 2028, USD Million
- TABLE 65 North America Soft Tissue Orthopedic Devices Market by Country, 2018 - 2021, USD Million
- TABLE 66 North America Soft Tissue Orthopedic Devices Market by Country, 2022 - 2028, USD Million
- TABLE 67 North America Foot And Ankle Devices Market by Country, 2018 - 2021, USD Million
- TABLE 68 North America Foot And Ankle Devices Market by Country, 2022 - 2028, USD Million
- TABLE 69 US Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 70 US Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 71 US Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 72 US Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 73 US Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 74 US Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 75 Canada Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 76 Canada Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 77 Canada Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 78 Canada Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 79 Canada Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 80 Canada Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 81 Mexico Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 82 Mexico Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 83 Mexico Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 84 Mexico Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 85 Mexico Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 86 Mexico Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 87 Rest of North America Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 88 Rest of North America Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 89 Rest of North America Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 90 Rest of North America Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 91 Rest of North America Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 92 Rest of North America Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 93 Europe Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 94 Europe Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 95 Europe Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 96 Europe Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 97 Europe Trauma Market by Country, 2018 - 2021, USD Million
- TABLE 98 Europe Trauma Market by Country, 2022 - 2028, USD Million
- TABLE 99 Europe Osteoarthritis Market by Country, 2018 - 2021, USD Million
- TABLE 100 Europe Osteoarthritis Market by Country, 2022 - 2028, USD Million
- TABLE 101 Europe Rheumatoid Arthritis Market by Country, 2018 - 2021, USD Million
- TABLE 102 Europe Rheumatoid Arthritis Market by Country, 2022 - 2028, USD Million
- TABLE 103 Europe Hammertoe Market by Country, 2018 - 2021, USD Million
- TABLE 104 Europe Hammertoe Market by Country, 2022 - 2028, USD Million
- TABLE 105 Europe Osteoporosis Market by Country, 2018 - 2021, USD Million
- TABLE 106 Europe Osteoporosis Market by Country, 2022 - 2028, USD Million
- TABLE 107 Europe Neurological Disorders Market by Country, 2018 - 2021, USD Million
- TABLE 108 Europe Neurological Disorders Market by Country, 2022 - 2028, USD Million
- TABLE 109 Europe Bunions Market by Country, 2018 - 2021, USD Million
- TABLE 110 Europe Bunions Market by Country, 2022 - 2028, USD Million
- TABLE 111 Europe Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 112 Europe Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 113 Europe Orthopedic Fixation Market by Country, 2018 - 2021, USD Million
- TABLE 114 Europe Orthopedic Fixation Market by Country, 2022 - 2028, USD Million
- TABLE 115 Europe Joint Implants Market by Country, 2018 - 2021, USD Million
- TABLE 116 Europe Joint Implants Market by Country, 2022 - 2028, USD Million
- TABLE 117 Europe Bracing & Support Market by Country, 2018 - 2021, USD Million
- TABLE 118 Europe Bracing & Support Market by Country, 2022 - 2028, USD Million
- TABLE 119 Europe Prosthetics Market by Country, 2018 - 2021, USD Million
- TABLE 120 Europe Prosthetics Market by Country, 2022 - 2028, USD Million
- TABLE 121 Europe Soft Tissue Orthopedic Devices Market by Country, 2018 - 2021, USD Million
- TABLE 122 Europe Soft Tissue Orthopedic Devices Market by Country, 2022 - 2028, USD Million
- TABLE 123 Europe Foot And Ankle Devices Market by Country, 2018 - 2021, USD Million
- TABLE 124 Europe Foot And Ankle Devices Market by Country, 2022 - 2028, USD Million
- TABLE 125 Germany Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 126 Germany Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 127 Germany Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 128 Germany Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 129 Germany Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 130 Germany Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 131 UK Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 132 UK Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 133 UK Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 134 UK Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 135 UK Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 136 UK Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 137 France Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 138 France Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 139 France Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 140 France Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 141 France Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 142 France Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 143 Russia Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 144 Russia Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 145 Russia Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 146 Russia Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 147 Russia Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 148 Russia Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 149 Spain Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 150 Spain Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 151 Spain Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 152 Spain Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 153 Spain Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 154 Spain Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 155 Italy Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 156 Italy Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 157 Italy Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 158 Italy Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 159 Italy Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 160 Italy Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 161 Rest of Europe Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 162 Rest of Europe Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 163 Rest of Europe Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 164 Rest of Europe Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 165 Rest of Europe Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 166 Rest of Europe Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 167 Asia Pacific Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 168 Asia Pacific Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 169 Asia Pacific Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 170 Asia Pacific Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 171 Asia Pacific Trauma Market by Country, 2018 - 2021, USD Million
- TABLE 172 Asia Pacific Trauma Market by Country, 2022 - 2028, USD Million
- TABLE 173 Asia Pacific Osteoarthritis Market by Country, 2018 - 2021, USD Million
- TABLE 174 Asia Pacific Osteoarthritis Market by Country, 2022 - 2028, USD Million
- TABLE 175 Asia Pacific Rheumatoid Arthritis Market by Country, 2018 - 2021, USD Million
- TABLE 176 Asia Pacific Rheumatoid Arthritis Market by Country, 2022 - 2028, USD Million
- TABLE 177 Asia Pacific Hammertoe Market by Country, 2018 - 2021, USD Million
- TABLE 178 Asia Pacific Hammertoe Market by Country, 2022 - 2028, USD Million
- TABLE 179 Asia Pacific Osteoporosis Market by Country, 2018 - 2021, USD Million
- TABLE 180 Asia Pacific Osteoporosis Market by Country, 2022 - 2028, USD Million
- TABLE 181 Asia Pacific Neurological Disorders Market by Country, 2018 - 2021, USD Million
- TABLE 182 Asia Pacific Neurological Disorders Market by Country, 2022 - 2028, USD Million
- TABLE 183 Asia Pacific Bunions Market by Country, 2018 - 2021, USD Million
- TABLE 184 Asia Pacific Bunions Market by Country, 2022 - 2028, USD Million
- TABLE 185 Asia Pacific Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 186 Asia Pacific Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 187 Asia Pacific Orthopedic Fixation Market by Country, 2018 - 2021, USD Million
- TABLE 188 Asia Pacific Orthopedic Fixation Market by Country, 2022 - 2028, USD Million
- TABLE 189 Asia Pacific Joint Implants Market by Country, 2018 - 2021, USD Million
- TABLE 190 Asia Pacific Joint Implants Market by Country, 2022 - 2028, USD Million
- TABLE 191 Asia Pacific Bracing & Support Market by Country, 2018 - 2021, USD Million
- TABLE 192 Asia Pacific Bracing & Support Market by Country, 2022 - 2028, USD Million
- TABLE 193 Asia Pacific Prosthetics Market by Country, 2018 - 2021, USD Million
- TABLE 194 Asia Pacific Prosthetics Market by Country, 2022 - 2028, USD Million
- TABLE 195 Asia Pacific Soft Tissue Orthopedic Devices Market by Country, 2018 - 2021, USD Million
- TABLE 196 Asia Pacific Soft Tissue Orthopedic Devices Market by Country, 2022 - 2028, USD Million
- TABLE 197 Asia Pacific Foot And Ankle Devices Market by Country, 2018 - 2021, USD Million
- TABLE 198 Asia Pacific Foot And Ankle Devices Market by Country, 2022 - 2028, USD Million
- TABLE 199 China Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 200 China Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 201 China Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 202 China Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 203 China Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 204 China Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 205 Japan Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 206 Japan Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 207 Japan Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 208 Japan Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 209 Japan Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 210 Japan Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 211 India Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 212 India Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 213 India Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 214 India Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 215 India Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 216 India Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 217 South Korea Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 218 South Korea Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 219 South Korea Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 220 South Korea Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 221 South Korea Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 222 South Korea Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 223 Singapore Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 224 Singapore Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 225 Singapore Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 226 Singapore Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 227 Singapore Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 228 Singapore Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 229 Malaysia Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 230 Malaysia Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 231 Malaysia Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 232 Malaysia Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 233 Malaysia Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 234 Malaysia Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 235 Rest of Asia Pacific Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 236 Rest of Asia Pacific Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 237 Rest of Asia Pacific Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 238 Rest of Asia Pacific Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 239 Rest of Asia Pacific Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 240 Rest of Asia Pacific Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 241 LAMEA Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 242 LAMEA Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 243 LAMEA Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 244 LAMEA Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 245 LAMEA Trauma Market by Country, 2018 - 2021, USD Million
- TABLE 246 LAMEA Trauma Market by Country, 2022 - 2028, USD Million
- TABLE 247 LAMEA Osteoarthritis Market by Country, 2018 - 2021, USD Million
- TABLE 248 LAMEA Osteoarthritis Market by Country, 2022 - 2028, USD Million
- TABLE 249 LAMEA Rheumatoid Arthritis Market by Country, 2018 - 2021, USD Million
- TABLE 250 LAMEA Rheumatoid Arthritis Market by Country, 2022 - 2028, USD Million
- TABLE 251 LAMEA Hammertoe Market by Country, 2018 - 2021, USD Million
- TABLE 252 LAMEA Hammertoe Market by Country, 2022 - 2028, USD Million
- TABLE 253 LAMEA Osteoporosis Market by Country, 2018 - 2021, USD Million
- TABLE 254 LAMEA Osteoporosis Market by Country, 2022 - 2028, USD Million
- TABLE 255 LAMEA Neurological Disorders Market by Country, 2018 - 2021, USD Million
- TABLE 256 LAMEA Neurological Disorders Market by Country, 2022 - 2028, USD Million
- TABLE 257 LAMEA Bunions Market by Country, 2018 - 2021, USD Million
- TABLE 258 LAMEA Bunions Market by Country, 2022 - 2028, USD Million
- TABLE 259 LAMEA Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 260 LAMEA Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 261 LAMEA Orthopedic Fixation Market by Country, 2018 - 2021, USD Million
- TABLE 262 LAMEA Orthopedic Fixation Market by Country, 2022 - 2028, USD Million
- TABLE 263 LAMEA Joint Implants Market by Country, 2018 - 2021, USD Million
- TABLE 264 LAMEA Joint Implants Market by Country, 2022 - 2028, USD Million
- TABLE 265 LAMEA Bracing & Support Market by Country, 2018 - 2021, USD Million
- TABLE 266 LAMEA Bracing & Support Market by Country, 2022 - 2028, USD Million
- TABLE 267 LAMEA Prosthetics Market by Country, 2018 - 2021, USD Million
- TABLE 268 LAMEA Prosthetics Market by Country, 2022 - 2028, USD Million
- TABLE 269 LAMEA Soft Tissue Orthopedic Devices Market by Country, 2018 - 2021, USD Million
- TABLE 270 LAMEA Soft Tissue Orthopedic Devices Market by Country, 2022 - 2028, USD Million
- TABLE 271 LAMEA Foot And Ankle Devices Market by Country, 2018 - 2021, USD Million
- TABLE 272 LAMEA Foot And Ankle Devices Market by Country, 2022 - 2028, USD Million
- TABLE 273 Brazil Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 274 Brazil Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 275 Brazil Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 276 Brazil Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 277 Brazil Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 278 Brazil Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 279 Argentina Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 280 Argentina Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 281 Argentina Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 282 Argentina Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 283 Argentina Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 284 Argentina Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 285 UAE Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 286 UAE Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 287 UAE Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 288 UAE Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 289 UAE Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 290 UAE Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 291 Saudi Arabia Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 292 Saudi Arabia Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 293 Saudi Arabia Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 294 Saudi Arabia Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 295 Saudi Arabia Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 296 Saudi Arabia Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 297 South Africa Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 298 South Africa Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 299 South Africa Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 300 South Africa Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 301 South Africa Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 302 South Africa Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 303 Nigeria Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 304 Nigeria Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 305 Nigeria Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 306 Nigeria Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 307 Nigeria Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 308 Nigeria Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 309 Rest of LAMEA Foot And Ankle Devices Market, 2018 - 2021, USD Million
- TABLE 310 Rest of LAMEA Foot And Ankle Devices Market, 2022 - 2028, USD Million
- TABLE 311 Rest of LAMEA Foot And Ankle Devices Market by Application, 2018 - 2021, USD Million
- TABLE 312 Rest of LAMEA Foot And Ankle Devices Market by Application, 2022 - 2028, USD Million
- TABLE 313 Rest of LAMEA Foot And Ankle Devices Market by Product, 2018 - 2021, USD Million
- TABLE 314 Rest of LAMEA Foot And Ankle Devices Market by Product, 2022 - 2028, USD Million
- TABLE 315 key Information - Stryker Corporation
- TABLE 316 Key Information - DePuy Synthes
- TABLE 317 key Information - Zimmer Biomet Holdings, Inc.
- TABLE 318 key Information - Smith & Nephew PLC
- TABLE 319 Key Information - Ossur Hf.
- TABLE 320 Key Information - Enovis Corporation
- TABLE 321 Key Information - Acumed LLC
- TABLE 322 Key information - Arthrex, Inc.
- TABLE 323 Key Information - Paragon 28, Inc.
- TABLE 324 Key Information - Orthofix Medical, Inc.
List of Figures
- FIG 1 Methodology for the research
- FIG 2 KBV Cardinal Matrix
- FIG 3 Key Leading Strategies: Percentage Distribution (2018-2022)
- FIG 4 Key Strategic Move: (Product Launches and Product Expansions: 2019, Feb - 2023, Jan) Leading Players
- FIG 5 Global Foot And Ankle Devices Market Share by Application, 2021
- FIG 6 Global Foot And Ankle Devices Market Share by Application, 2028
- FIG 7 Global Foot And Ankle Devices Market by Application, 2018 - 2028, USD Million
- FIG 8 Global Foot And Ankle Devices Market Share by Product, 2021
- FIG 9 Global Foot And Ankle Devices Market Share by Product, 2028
- FIG 10 Global Foot And Ankle Devices Market by Product, 2018 - 2028, USD Million
- FIG 11 Global Foot And Ankle Devices Market Share by Region, 2021
- FIG 12 Global Foot And Ankle Devices Market Share by Region, 2028
- FIG 13 Global Foot And Ankle Devices Market by Region, 2018 - 2028, USD Million
- FIG 14 Recent strategies and developments: Stryker Corporation
- FIG 15 Recent strategies and developments: Zimmer Biomet Holdings, Inc.
- FIG 16 Recent strategies and developments: Smith & Nephew PLC
- FIG 17 Recent strategies and developments: Ossur Hf.
- FIG 18 Recent strategies and developments: Enovis Corporation
- FIG 19 Recent strategies and developments: Paragon 28, Inc.
- FIG 20 Recent strategies and developments: Orthofix Medical, Inc.
The Global Foot And Ankle Devices Market size is expected to reach $6.1 billion by 2028, rising at a market growth of 7.1% CAGR during the forecast period.
Many foot and ankle fractures brought on by ailments, including arthritis, diabetes, hammertoes, bunions, and other orthopedic illnesses, can be treated with foot and ankle devices. These medical tools can also replace an amputated or lost foot due to an accident, trauma, congenital abnormality, or any other reason. The foot and ankle devices comprise tools for joint replacement, arthrodesis, and internal fixation treatments that aid in ligament repair and restoration.
These devices assist weak muscles and lessen, stop, or restrict foot and lower leg motion to improve movement patterns. They are also used to maintain joint alignment, adapt deformity, and decrease spasticity. In order to protect the leg and foot, these devices are worn with socks and shoes. Such devices come in several shapes and materials, particularly if they are being developed for children and youngsters, they can be modified for use and growth. Since they are composed of thermoformed plastic, tweaks and changes are possible.
Rheumatoid arthritis (RA) affects roughly one percent of people worldwide, with women being affected at least twice as often as males. More than 23 million people are thought to have RA, according to the World Health Organization. Unfortunately, the way that patients with RA are currently receiving healthcare is not equipped to handle the predicted financial burden of the disease in the coming years. By 2030, the number of RA sufferers could double, according to experts. This will be due to the transition of the baby boom generation between age groups, a greater understanding of RA among professionals, and a somewhat increased incidence of RA is present in people over the age of 70.
Additionally, over 200 million people worldwide are thought to have osteoporosis. Recent data from the International Osteoporosis Foundation show that 1 in 3 women over 50 and 1 in 5 men will sustain an osteoporotic fracture during their lifetimes. Moreover, osteoporosis reduces life expectancy, lengthens lifespan after adjusting for disability, and places a significant financial load on the health insurance systems of the nations in charge of providing care for such individuals. The market for foot and ankle devices is therefore anticipated to expand rapidly.
COVID-19 Impact Analysis
Foot and ankle trauma patients decreased significantly during the pandemic. As a result, the need for foot and ankle devices decreased as hospitals turned their attention to the COVID-19 outbreak. As a result of being unable to focus on routine patients, orthopedics professionals suffered. The treatment of COVID-19 patients required the redeployment of medical personnel. The absence of hospital employees and operating infrastructure slowed market expansion. With constrained supply chains hindering the distribution of these devices and the decrease in treatments worldwide, the COVID-19 pandemic severely impacted the foot and ankle devices market.
Market Growth Factors
Rising foot and ankle issues worldwide
Rheumatoid arthritis, osteoarthritis, and posttraumatic arthritis are the three main kinds of arthritis commonly affecting the foot and ankle. Mobility may be increased using a brace, like an ankle-foot orthosis (AFO). Moreover, using orthotics or specially constructed braces helps lessen pain and reduce pressure on the foot. Compared with standard goods, benefits, including increased efficacy, lower cost, more patient comfort, and convenience of use, are anticipated to increase the demand for these devices and thus benefit the foot and ankle devices market.
Increasing incorporation of technology and strategic initiatives
Patient volume, and surgical expertise, nations like China have also demonstrated vital indicators. The sector for 3D printed orthopedic devices is characterized by the inclusion of individualized prosthetics and metallic implants made of cobalt-chromium alloy, stainless steel, and alpha-beta titanium alloy (Ti-6Al-4V). Therefore, the strategic initiatives by key market participants and the higher acceptance of advanced technologies in the manufacturing of devices are propelling the expansion of the market.
Market Restraining Factors
Severe side effects of foot and ankle devices
Long-term usage of a brace can result in muscular atrophy and deconditioning of the ligaments, joints, and surrounding muscles. This is especially true if daily activities to reinforce the area are skipped. A decline in a joint's functional level, increased pain and discomfort in the joint without the support brace, and greater dependence on the brace for daily activities are all warning indications of wearing a brace for an excessively long time. These could be obstacles to the overall expansion of the foot and ankle devices market.
Product Outlook
Based on product, the foot and ankle devices market is categorized into bracing & support, joint implants, soft tissue orthopedic devices, orthopedic fixation, and prosthetics. The bracing and supporting segment procured a considerable growth rate in the foot and ankle devices market in 2021. The rise of the segment is owed to the benefits of these devices. Devices worn around the foot and ankle, such as braces and supports, help to stabilize the joint by offering support. Injuries, including sprains, fractures, and tendinitis, are frequently treated with them. When a person is at risk of acquiring foot and ankle issues, braces can also be used to avoid injury.
Application Outlook
On the basis of application, the foot and ankle devices market is divided into hammertoe, trauma, osteoarthritis, rheumatoid arthritis, neurological disorders, bunions, and osteoporosis. The osteoporosis segment garnered a remarkable growth rate in the foot and ankle devices market in 2021. Expansion of the segment is owed to the rising demand for support devices in most osteoporosis cases. Osteoporosis could predispose to particular ankle fractures, a mechanically tenable assumption. According to extensive epidemiological research, both men and women with high BMI have lower risks of fragility fractures and greater BMD.
Regional Outlook
Region wise, the foot and ankle devices market is analyzed across North America, Europe, Asia Pacific, and LAMEA. The North America region acquired the highest revenue share in the foot and ankle devices market in 2021. The prevalence of a sizable senior population pool and the high frequency of sports and physical activity-related injuries are the primary causes of this region's big proportion. Almost 55.6 million adults in the US, or roughly 16.9% of the population, are 65 years of age or older, according to America's Health Rankings Senior Report.
The major strategies followed by the market participants are Product Launches. Based on the Analysis presented in the Cardinal matrix; DePuy Synthes (Johnson & Johnson) is the major forerunner in sthe Foot and Ankle Devices Market. Companies such as Enovis Corporation, Stryker Corporation, and Paragon 28, Inc. are some of the key innovators in Foot and Ankle Devices Market.
The market research report covers the analysis of key stake holders of the market. Key companies profiled in the report include Stryker Corporation, DePuy Synthes (Johnson & Johnson), Zimmer Biomet Holdings, Inc., Smith & Nephew PLC, Ossur Hf., Enovis Corporation, Acumed LLC (Colson Medical, Inc.), Arthrex, Inc., Paragon 28, Inc. and Orthofix Medical, Inc.
Recent strategies deployed in Foot and Ankle Devices Market
Partnerships, Collaborations, and Agreements:
Jul-2022: Zimmer Biomet signed a three-year agreement with the Hospital for Special Surgery (HSS) for opening the HSS/Zimmer Biomet Innovation Center for Artificial Intelligence (AI) in Robotic Joint Replacement. The companies want to create new decision-support systems, powered by machine learning and data collection, for providing data-driven advice for robotic joint surgery, to surgeons. A team of HSS surgeons and ZB data scientists will work together throughout the agreement to create and test hypotheses that employ data-driven insights to address typical clinical difficulties related to joint replacement. The objective is to inform the creation of algorithms for decision support tools using the validated hypotheses.
Apr-2021: Zimmer Biomet signed a distribution agreement with Device Technologies for the distribution of its surgical product portfolio, with trauma and foot & ankle portfolio, and sports medicine in New Zealand and Australia.
Acquisition and Mergers:
Jan-2023: Zimmer Biomet Holdings signed an agreement to acquire Embody, Inc.., a medical device company engaged in soft tissue healing. The acquisition would broaden Zimmer's portfolio for providing support in the healing of the most difficult orthopedic soft tissue injuries.
Feb-2022: DePuy Synthes completed the acquisition of CrossRoads Extremity Systems, a foot and ankle company that focuses on providing a wide range of procedure-scientific, sterile packed instrumentation and implant systems for lower extremity indications. The acquisition broadened the company's foot and ankle procedure portfolio.
Jan-2022: Stryker is acquiring Vocera Communications, a company engaged in the development of clinical workflow and communication technologies. The acquisition would strengthen Stryker's innovative and highly complementary portfolio of tools, for connecting caregivers and disparate data-generating medical devices, with the aim to improve provider workflow and boost patient safety outcomes.
Jan-2022: Paragon 28, Inc. completed the acquisition of Disior Oy, a three-dimensional analytics pre-operative planning software company focused on complex ankle and foot anatomy. The acquisition accelerated the internal research and development efforts of Paragon and delivers unique pre-operative planning capabilities to surgeons.
Dec-2021: DePuy Synthes acquired OrthoSpin, Ltd., a developer and manufacturer of an automated strut system, a first-of-its-kind technology, which is used together with DePuy Synthes MAXFRAME™ Multi-Axial Correction System, an external ring fixation system. The acquisition enabled DePuy to better serve patients and healthcare professionals at the time of the post-complex surgery recovery period.
Jul-2021: Enovis acquired Mathys AG Bettlach, a company engaged in developing, producing, and distributing implants for artificial joint replacement. The acquisition strengthened DJO's patient-focused portfolio and expanded its reconstructive portfolio.
Jun-2021: Paragon 28 acquired Additive Orthopaedics' product lines including its FDA-approved, first in the world 3D printed Specific Talus Spacer utilized for the treatment of Avascular Necrosis for Humanitarian use. The acquisition provided customers exclusive access to Paragon the only FDA-approved patient-specific total talus replacement implant.
Jan-2021: Smith & Nephew took over Integra LifeSciences Holdings Corporation's Extremity Orthopaedics business. The acquisition assists the former company's strategy of investing in a higher growth strategy and expanding the company's extremities business.
Product Launches and Product Expansions:
Jan-2023: Enovis Corporation unveiled the DynaClip Quattro and DynaClip Delta bone staples, the new addition to Enovis' DynaClip family of bone fixation systems. These staples are pre-loaded on disposable inserters that ease quick deployment and correct leg positioning. Additionally, DynaClip Delta staples are created for offering easier and faster first metatarsophalangeal (MTP) fusion procedures without compromising on performance and durability.
Dec-2022: Stryker unveiled a suture anchor system for ankle and foot procedures, Citrefix. This is a disposable suture anchor system that has a resorbable material anchor body, made with Citregen, an elastomeric material made from a citrate polymer specifically made to mimic bone chemistry for controlled resorption without chronic inflammation.
Nov-2022: Enovis released the DynaNail Helix, a new addition to the DynaNail family and the company's foot and ankle product portfolio. DynaNail Helix is developed for achieving dynamic compression for subtalar fusion with an anatomically, innovative, friendly design, and simple screw-like insertion.
Sep-2022: Stryker introduced the Pulse Intelligent Delivery Platform, a complete circle product, and fulfillment offering developed for high-volume ankle and foot procedures in ambulatory surgery center setups. This platform offers an integrated product delivery and configuration offering for procedures through smart configurations, personalized inventory, and automated services.
Jul-2022: Arthrex unveiled the Arthrex Minimally Invasive Bunionectomy System for providing an alternative solution to traditional bunion surgery. This system is being designed to be an all-inclusive set for facilitating minimally invasive surgical bunion correction. It is clinically proven to get the same or better results as compared to traditional surgery with less swelling, downtime, and pain.
Jun-2022: Enovis announced the release of the Arsenal Ankle Plating System, comprising 37 anatomically designed plates across nine plate families, providing a plating solution for an ankle fracture. The Arsenal Ankle plating System came up with a variable angle locking technology. It allows a cone angulation of 60°, with a multi-lock function, which accepts repeatable lock function and relocking of screw for plate interface. This offers a powerful combination of patented and innovative solutions which would address all fracture personalities.
Apr-2022: Paragon 28, Inc. announced the launch of the R3ACT Stabilization system, a simple solution that helps in multi-stage soft tissue healing after chronic or acute syndesmotic injury to the ankle.
Mar-2022: Acumed launched Ankle Syndesmosis Repair System with Acu-Sinch Knotless technology. Acu-Sinch Knotless Implant allows the dynamic stabilization of laxity or syndesmotic disruptions to the tibiofibular joint. Ankle Syndesmosis Repair System offers the user control to place the medical button subcutaneously without the requirement for direct visualization. Without the necessity for direct visualization, the release mechanism offers the user control to implant the medial button subcutaneously. The Flip Button position may be seen under fluoroscopy because of radiographic indications and the trigger mechanism's tactile feedback. The Acumed and OsteoMed fibula fracture fixation plates as well as intramedullary nails with 3.5 mm nonlocking screw holes can all be used in conjunction with the Acu-Sinch Knotless buttons, either alone or in addition to a washer.
Scope of the Study
Market Segments covered in the Report:
By Application
- Trauma
- Osteoarthritis
- Rheumatoid Arthritis
- Hammertoe
- Osteoporosis
- Neurological Disorders
- Bunions
By Product
- Orthopedic Fixation
- Joint Implants
- Bracing & Support
- Prosthetics
- Soft Tissue Orthopedic Devices
By Geography
- North America
- US
- Canada
- Mexico
- Rest of North America
- Europe
- Germany
- UK
- France
- Russia
- Spain
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Singapore
- Malaysia
- Rest of Asia Pacific
- LAMEA
- Brazil
- Argentina
- UAE
- Saudi Arabia
- South Africa
- Nigeria
- Rest of LAMEA
Companies Profiled
- Stryker Corporation
- DePuy Synthes (Johnson & Johnson)
- Zimmer Biomet Holdings, Inc.
- Smith & Nephew PLC
- Ossur Hf.
- Enovis Corporation
- Acumed LLC (Colson Medical, Inc.)
- Arthrex, Inc.
- Paragon 28, Inc.
- Orthofix Medical, Inc.
Unique Offerings from KBV Research
- Exhaustive coverage
- Highest number of market tables and figures
- Subscription based model available
- Guaranteed best price
- Assured post sales research support with 10% customization free
Table of Contents
Chapter 1. Market Scope & Methodology
- 1.1 Market Definition
- 1.2 Objectives
- 1.3 Market Scope
- 1.4 Segmentation
- 1.4.1 Global Foot And Ankle Devices Market, by Application
- 1.4.2 Global Foot And Ankle Devices Market, by Product
- 1.4.3 Global Foot And Ankle Devices Market, by Geography
- 1.5 Methodology for the research
Chapter 2. Market Overview
- 2.1 Introduction
- 2.1.1 Overview
- 2.1.1.1 Market Composition & Scenario
- 2.1.1 Overview
- 2.2 Key Factors Impacting the Market
- 2.2.1 Market Drivers
- 2.2.2 Market Restraints
Chapter 3. Competition Analysis - Global
- 3.1 KBV Cardinal Matrix
- 3.2 Recent Industry Wide Strategic Developments
- 3.2.1 Partnerships, Collaborations and Agreements
- 3.2.2 Product Launches and Product Expansions
- 3.2.3 Acquisition and Mergers
- 3.2.4 Approvals & Trials
- 3.3 Top Winning Strategies
- 3.3.1 Key Leading Strategies: Percentage Distribution (2018-2022)
- 3.3.2 Key Strategic Move: (Product Launches and Product Expansions: 2019, Feb - 2023, Jan) Leading Players
Chapter 4. Global Foot And Ankle Devices Market by Application
- 4.1 Global Trauma Market by Region
- 4.2 Global Osteoarthritis Market by Region
- 4.3 Global Rheumatoid Arthritis Market by Region
- 4.4 Global Hammertoe Market by Region
- 4.5 Global Osteoporosis Market by Region
- 4.6 Global Neurological Disorders Market by Region
- 4.7 Global Bunions Market by Region
Chapter 5. Global Foot And Ankle Devices Market by Product
- 5.1 Global Orthopedic Fixation Market by Region
- 5.2 Global Joint Implants Market by Region
- 5.3 Global Bracing & Support Market by Region
- 5.4 Global Prosthetics Market by Region
- 5.5 Global Soft Tissue Orthopedic Devices Market by Region
Chapter 6. Global Foot And Ankle Devices Market by Region
- 6.1 North America Foot And Ankle Devices Market
- 6.1.1 North America Foot And Ankle Devices Market by Application
- 6.1.1.1 North America Trauma Market by Country
- 6.1.1.2 North America Osteoarthritis Market by Country
- 6.1.1.3 North America Rheumatoid Arthritis Market by Country
- 6.1.1.4 North America Hammertoe Market by Country
- 6.1.1.5 North America Osteoporosis Market by Country
- 6.1.1.6 North America Neurological Disorders Market by Country
- 6.1.1.7 North America Bunions Market by Country
- 6.1.2 North America Foot And Ankle Devices Market by Product
- 6.1.2.1 North America Orthopedic Fixation Market by Country
- 6.1.2.2 North America Joint Implants Market by Country
- 6.1.2.3 North America Bracing & Support Market by Country
- 6.1.2.4 North America Prosthetics Market by Country
- 6.1.2.5 North America Soft Tissue Orthopedic Devices Market by Country
- 6.1.3 North America Foot And Ankle Devices Market by Country
- 6.1.3.1 US Foot And Ankle Devices Market
- 6.1.3.1.1 US Foot And Ankle Devices Market by Application
- 6.1.3.1.2 US Foot And Ankle Devices Market by Product
- 6.1.3.2 Canada Foot And Ankle Devices Market
- 6.1.3.2.1 Canada Foot And Ankle Devices Market by Application
- 6.1.3.2.2 Canada Foot And Ankle Devices Market by Product
- 6.1.3.3 Mexico Foot And Ankle Devices Market
- 6.1.3.3.1 Mexico Foot And Ankle Devices Market by Application
- 6.1.3.3.2 Mexico Foot And Ankle Devices Market by Product
- 6.1.3.4 Rest of North America Foot And Ankle Devices Market
- 6.1.3.4.1 Rest of North America Foot And Ankle Devices Market by Application
- 6.1.3.4.2 Rest of North America Foot And Ankle Devices Market by Product
- 6.1.3.1 US Foot And Ankle Devices Market
- 6.1.1 North America Foot And Ankle Devices Market by Application
- 6.2 Europe Foot And Ankle Devices Market
- 6.2.1 Europe Foot And Ankle Devices Market by Application
- 6.2.1.1 Europe Trauma Market by Country
- 6.2.1.2 Europe Osteoarthritis Market by Country
- 6.2.1.3 Europe Rheumatoid Arthritis Market by Country
- 6.2.1.4 Europe Hammertoe Market by Country
- 6.2.1.5 Europe Osteoporosis Market by Country
- 6.2.1.6 Europe Neurological Disorders Market by Country
- 6.2.1.7 Europe Bunions Market by Country
- 6.2.2 Europe Foot And Ankle Devices Market by Product
- 6.2.2.1 Europe Orthopedic Fixation Market by Country
- 6.2.2.2 Europe Joint Implants Market by Country
- 6.2.2.3 Europe Bracing & Support Market by Country
- 6.2.2.4 Europe Prosthetics Market by Country
- 6.2.2.5 Europe Soft Tissue Orthopedic Devices Market by Country
- 6.2.3 Europe Foot And Ankle Devices Market by Country
- 6.2.3.1 Germany Foot And Ankle Devices Market
- 6.2.3.1.1 Germany Foot And Ankle Devices Market by Application
- 6.2.3.1.2 Germany Foot And Ankle Devices Market by Product
- 6.2.3.2 UK Foot And Ankle Devices Market
- 6.2.3.2.1 UK Foot And Ankle Devices Market by Application
- 6.2.3.2.2 UK Foot And Ankle Devices Market by Product
- 6.2.3.3 France Foot And Ankle Devices Market
- 6.2.3.3.1 France Foot And Ankle Devices Market by Application
- 6.2.3.3.2 France Foot And Ankle Devices Market by Product
- 6.2.3.4 Russia Foot And Ankle Devices Market
- 6.2.3.4.1 Russia Foot And Ankle Devices Market by Application
- 6.2.3.4.2 Russia Foot And Ankle Devices Market by Product
- 6.2.3.5 Spain Foot And Ankle Devices Market
- 6.2.3.5.1 Spain Foot And Ankle Devices Market by Application
- 6.2.3.5.2 Spain Foot And Ankle Devices Market by Product
- 6.2.3.6 Italy Foot And Ankle Devices Market
- 6.2.3.6.1 Italy Foot And Ankle Devices Market by Application
- 6.2.3.6.2 Italy Foot And Ankle Devices Market by Product
- 6.2.3.7 Rest of Europe Foot And Ankle Devices Market
- 6.2.3.7.1 Rest of Europe Foot And Ankle Devices Market by Application
- 6.2.3.7.2 Rest of Europe Foot And Ankle Devices Market by Product
- 6.2.3.1 Germany Foot And Ankle Devices Market
- 6.2.1 Europe Foot And Ankle Devices Market by Application
- 6.3 Asia Pacific Foot And Ankle Devices Market
- 6.3.1 Asia Pacific Foot And Ankle Devices Market by Application
- 6.3.1.1 Asia Pacific Trauma Market by Country
- 6.3.1.2 Asia Pacific Osteoarthritis Market by Country
- 6.3.1.3 Asia Pacific Rheumatoid Arthritis Market by Country
- 6.3.1.4 Asia Pacific Hammertoe Market by Country
- 6.3.1.5 Asia Pacific Osteoporosis Market by Country
- 6.3.1.6 Asia Pacific Neurological Disorders Market by Country
- 6.3.1.7 Asia Pacific Bunions Market by Country
- 6.3.2 Asia Pacific Foot And Ankle Devices Market by Product
- 6.3.2.1 Asia Pacific Orthopedic Fixation Market by Country
- 6.3.2.2 Asia Pacific Joint Implants Market by Country
- 6.3.2.3 Asia Pacific Bracing & Support Market by Country
- 6.3.2.4 Asia Pacific Prosthetics Market by Country
- 6.3.2.5 Asia Pacific Soft Tissue Orthopedic Devices Market by Country
- 6.3.3 Asia Pacific Foot And Ankle Devices Market by Country
- 6.3.3.1 China Foot And Ankle Devices Market
- 6.3.3.1.1 China Foot And Ankle Devices Market by Application
- 6.3.3.1.2 China Foot And Ankle Devices Market by Product
- 6.3.3.2 Japan Foot And Ankle Devices Market
- 6.3.3.2.1 Japan Foot And Ankle Devices Market by Application
- 6.3.3.2.2 Japan Foot And Ankle Devices Market by Product
- 6.3.3.3 India Foot And Ankle Devices Market
- 6.3.3.3.1 India Foot And Ankle Devices Market by Application
- 6.3.3.3.2 India Foot And Ankle Devices Market by Product
- 6.3.3.4 South Korea Foot And Ankle Devices Market
- 6.3.3.4.1 South Korea Foot And Ankle Devices Market by Application
- 6.3.3.4.2 South Korea Foot And Ankle Devices Market by Product
- 6.3.3.5 Singapore Foot And Ankle Devices Market
- 6.3.3.5.1 Singapore Foot And Ankle Devices Market by Application
- 6.3.3.5.2 Singapore Foot And Ankle Devices Market by Product
- 6.3.3.6 Malaysia Foot And Ankle Devices Market
- 6.3.3.6.1 Malaysia Foot And Ankle Devices Market by Application
- 6.3.3.6.2 Malaysia Foot And Ankle Devices Market by Product
- 6.3.3.7 Rest of Asia Pacific Foot And Ankle Devices Market
- 6.3.3.7.1 Rest of Asia Pacific Foot And Ankle Devices Market by Application
- 6.3.3.7.2 Rest of Asia Pacific Foot And Ankle Devices Market by Product
- 6.3.3.1 China Foot And Ankle Devices Market
- 6.3.1 Asia Pacific Foot And Ankle Devices Market by Application
- 6.4 LAMEA Foot And Ankle Devices Market
- 6.4.1 LAMEA Foot And Ankle Devices Market by Application
- 6.4.1.1 LAMEA Trauma Market by Country
- 6.4.1.2 LAMEA Osteoarthritis Market by Country
- 6.4.1.3 LAMEA Rheumatoid Arthritis Market by Country
- 6.4.1.4 LAMEA Hammertoe Market by Country
- 6.4.1.5 LAMEA Osteoporosis Market by Country
- 6.4.1.6 LAMEA Neurological Disorders Market by Country
- 6.4.1.7 LAMEA Bunions Market by Country
- 6.4.2 LAMEA Foot And Ankle Devices Market by Product
- 6.4.2.1 LAMEA Orthopedic Fixation Market by Country
- 6.4.2.2 LAMEA Joint Implants Market by Country
- 6.4.2.3 LAMEA Bracing & Support Market by Country
- 6.4.2.4 LAMEA Prosthetics Market by Country
- 6.4.2.5 LAMEA Soft Tissue Orthopedic Devices Market by Country
- 6.4.3 LAMEA Foot And Ankle Devices Market by Country
- 6.4.3.1 Brazil Foot And Ankle Devices Market
- 6.4.3.1.1 Brazil Foot And Ankle Devices Market by Application
- 6.4.3.1.2 Brazil Foot And Ankle Devices Market by Product
- 6.4.3.2 Argentina Foot And Ankle Devices Market
- 6.4.3.2.1 Argentina Foot And Ankle Devices Market by Application
- 6.4.3.2.2 Argentina Foot And Ankle Devices Market by Product
- 6.4.3.3 UAE Foot And Ankle Devices Market
- 6.4.3.3.1 UAE Foot And Ankle Devices Market by Application
- 6.4.3.3.2 UAE Foot And Ankle Devices Market by Product
- 6.4.3.4 Saudi Arabia Foot And Ankle Devices Market
- 6.4.3.4.1 Saudi Arabia Foot And Ankle Devices Market by Application
- 6.4.3.4.2 Saudi Arabia Foot And Ankle Devices Market by Product
- 6.4.3.5 South Africa Foot And Ankle Devices Market
- 6.4.3.5.1 South Africa Foot And Ankle Devices Market by Application
- 6.4.3.5.2 South Africa Foot And Ankle Devices Market by Product
- 6.4.3.6 Nigeria Foot And Ankle Devices Market
- 6.4.3.6.1 Nigeria Foot And Ankle Devices Market by Application
- 6.4.3.6.2 Nigeria Foot And Ankle Devices Market by Product
- 6.4.3.7 Rest of LAMEA Foot And Ankle Devices Market
- 6.4.3.7.1 Rest of LAMEA Foot And Ankle Devices Market by Application
- 6.4.3.7.2 Rest of LAMEA Foot And Ankle Devices Market by Product
- 6.4.3.1 Brazil Foot And Ankle Devices Market
- 6.4.1 LAMEA Foot And Ankle Devices Market by Application
Chapter 7. Company Profiles
- 7.1 Stryker Corporation
- 7.1.1 Company Overview
- 7.1.2 Financial Analysis
- 7.1.3 Segmental and Regional Analysis
- 7.1.4 Research & Development Expense
- 7.1.5 Recent strategies and developments:
- 7.1.5.1 Product Launches and Product Expansions:
- 7.1.5.2 Acquisition and Mergers:
- 7.2 DePuy Synthes (Johnson & Johnson)
- 7.2.1 Company Overview
- 7.2.2 Financial Analysis
- 7.2.3 Segmental &Regional Analysis
- 7.2.4 Research & Development Expenses
- 7.2.5 Recent strategies and developments:
- 7.2.5.1 Acquisition and Mergers:
- 7.3 Zimmer Biomet Holdings, Inc.
- 7.3.1 Company Overview
- 7.3.2 Financial Analysis
- 7.3.3 Regional Analysis
- 7.3.4 Research & Development Expense
- 7.3.5 Recent strategies and developments:
- 7.3.5.1 Partnerships, Collaborations, and Agreements:
- 7.3.5.2 Product Launches and Product Expansions:
- 7.3.5.3 Acquisition and Mergers:
- 7.4 Smith & Nephew PLC
- 7.4.1 Company Overview
- 7.4.2 Financial Analysis
- 7.4.3 Regional and Segmental Analysis
- 7.4.4 Research & Development Expense
- 7.4.5 Recent strategies and developments:
- 7.4.5.1 Product Launches and Product Expansions:
- 7.4.5.2 Acquisition and Mergers:
- 7.5 Ossur Hf.
- 7.5.1 Company Overview
- 7.5.2 Financial Analysis
- 7.5.3 Segmental and Regional Analysis
- 7.5.4 Research & Development Expense
- 7.5.5 Recent strategies and developments:
- 7.5.5.1 Product Launches and Product Expansions:
- 7.5.5.2 Acquisition and Mergers:
- 7.6 Enovis Corporation
- 7.6.1 Company Overview
- 7.6.2 Financial Analysis
- 7.6.3 Segmental and Regional Analysis
- 7.6.4 Research & Development Expenses
- 7.6.5 Recent strategies and developments:
- 7.6.5.1 Product Launches and Product Expansions:
- 7.6.5.2 Acquisition and Mergers:
- 7.6.5.3 Approvals & Trials:
- 7.7 Acumed LLC (Colson Medical, Inc.)
- 7.7.1 Company Overview
- 7.7.2 Recent strategies and developments:
- 7.7.2.1 Product Launches and Product Expansions:
- 7.8 Arthrex, Inc.
- 7.8.1 Company Overview
- 7.8.2 Recent strategies and developments:
- 7.8.2.1 Product Launches and Product Expansions:
- 7.9 Paragon 28, Inc.
- 7.9.1 Company Overview
- 7.9.2 Financial Analysis
- 7.9.3 Regional Analysis
- 7.9.4 Research & Development Expenses
- 7.9.5 Recent strategies and developments:
- 7.9.5.1 Product Launches and Product Expansions:
- 7.9.5.2 Acquisition and Mergers:
- 7.10. Orthofix Medical, Inc.
- 7.10.1 Company Overview
- 7.10.2 Financial Analysis
- 7.10.3 Segmental and Regional Analysis
- 7.10.4 Research & Development Expenses
- 7.10.5 Recent strategies and developments:
- 7.10.5.1 Product Launches and Product Expansions:
- 7.10.5.2 Approvals & Trials: