|
|
市場調査レポート
商品コード
1502226
インフルエンザ診断薬の世界市場 2024-2028Global influenza diagnostics market 2024-2028 |
||||||
カスタマイズ可能
|
|||||||
| インフルエンザ診断薬の世界市場 2024-2028 |
|
出版日: 2024年05月20日
発行: TechNavio
ページ情報: 英文 180 Pages
納期: 即納可能
|
全表示
- 概要
- 図表
- 目次
インフルエンザ診断薬市場は2023-2028年に5億1,750万米ドル、予測期間中のCAGRは7.22%で成長すると予測されます。
当レポートでは、インフルエンザ診断薬市場の全体的な分析、市場規模・予測、動向、成長促進要因、課題、約25のベンダーを網羅したベンダー分析などを掲載しています。
現在の市場シナリオ、最新動向と促進要因、市場環境全体に関する最新分析を提供しています。市場は、インフルエンザ罹患率の上昇、分子診断薬に対する需要の高まり、政府の支援策や資金援助によって牽引されています。
| 市場範囲 | |
|---|---|
| 基準年 | 2024 |
| 終了年 | 2028 |
| 予測期間 | 2024-2028 |
| 成長モメンタム | 加速 |
| 前年比2024年 | 6.6% |
| CAGR | 7.22% |
| 増分額 | 5億1,750万米ドル |
本調査では、今後数年間のインフルエンザ診断薬市場成長を牽引する主な要因の1つとして、インフルエンザ診断薬の承認件数増加を挙げています。また、インフルエンザウイルスの遺伝子特性解析研究の急増や、医師事務所検査室における迅速診断製品の採用増加も、市場の大きな需要につながります。
目次
第1章 エグゼクティブサマリー
- 市場概要
第2章 市場情勢
- 市場エコシステム
- 市場の特徴
- バリューチェーン分析
第3章 市場規模の評価
- 市場の定義
- 市場セグメント分析
- 市場規模 2023
- 市場の見通し 2023-2028
第4章 市場規模実績
- 世界インフルエンザ診断薬市場 2018-2022
- エンドユーザー別セグメント分析 2018-2022
- タイプ別セグメント分析 2018-2022
- 地域別セグメント分析 2018-2022
- 国別セグメント分析 2018-2022
第5章 ファイブフォース分析
- ファイブフォースの要約
- 買い手の交渉力
- 供給企業の交渉力
- 新規参入業者の脅威
- 代替品の脅威
- 競争の脅威
- 市況
第6章 市場セグメンテーション:エンドユーザー別
- 市場セグメンテーション
- 比較:エンドユーザー別
- 病院および診断研究所:市場規模と予測 2023-2028
- 学術研究センター:市場規模と予測 2023-2028
- 在宅ケア:市場規模と予測 2023-2028
- 市場機会:エンドユーザー別
第7章 市場セグメンテーション:タイプ別
- 市場セグメンテーション
- 比較:タイプ別
- ポイントオブケア検査:市場規模と予測 2023-2028
- 免疫診断:市場規模と予測 2023-2028
- 分子診断:市場規模と予測 2023-2028
- 市場機会:タイプ別
第8章 顧客情勢
- 顧客情勢の概要
第9章 地域別情勢
- 地域別セグメンテーション
- 地域別比較
- 北米:市場規模と予測 2023-2028
- 欧州:市場規模と予測 2023-2028
- アジア:市場規模と予測 2023-2028
- 世界のその他の地域:市場規模と予測 2023-2028
- 米国:市場規模と予測 2023-2028
- 中国:市場規模と予測 2023-2028
- ドイツ:市場規模と予測 2023-2028
- フランス:市場規模と予測 2023-2028
- カナダ:市場規模と予測 2023-2028
- 市場機会:地域別
第10章 促進要因・課題・機会・抑制要因
- 市場促進要因
- 市場の課題
- 促進要因と課題の影響
- 市場の機会・抑制要因
第11章 競合情勢
- 概要
- 競合情勢
- 混乱の状況
- 業界のリスク
第12章 競合分析
- 企業プロファイル
- 企業の市場ポジショニング
- Abbott Laboratories
- Becton Dickinson and Co.
- BICO Group AB
- Bio Rad Laboratories Inc.
- Biocartis NV
- bioMerieux SA
- Danaher Corp.
- DiaSorin SpA
- F. Hoffmann La Roche Ltd.
- Hologic Inc.
- Princeton BioMeditech Corp.
- Quidelortho Corp.
- Sd Biosensor Inc.
- Siemens Healthineers AG
- Thermo Fisher Scientific Inc.
第13章 付録
Exhibits:
- Exhibits1: Executive Summary - Chart on Market Overview
- Exhibits2: Executive Summary - Data Table on Market Overview
- Exhibits3: Executive Summary - Chart on Global Market Characteristics
- Exhibits4: Executive Summary - Chart on Market by Geography
- Exhibits5: Executive Summary - Chart on Market Segmentation by End-user
- Exhibits6: Executive Summary - Chart on Market Segmentation by Type
- Exhibits7: Executive Summary - Chart on Incremental Growth
- Exhibits8: Executive Summary - Data Table on Incremental Growth
- Exhibits9: Executive Summary - Chart on Company Market Positioning
- Exhibits10: Parent Market
- Exhibits11: Data Table on - Parent Market
- Exhibits12: Market characteristics analysis
- Exhibits13: Value Chain Analysis
- Exhibits14: Offerings of companies included in the market definition
- Exhibits15: Market segments
- Exhibits16: Chart on Global - Market size and forecast 2023-2028 ($ million)
- Exhibits17: Data Table on Global - Market size and forecast 2023-2028 ($ million)
- Exhibits18: Chart on Global Market: Year-over-year growth 2023-2028 (%)
- Exhibits19: Data Table on Global Market: Year-over-year growth 2023-2028 (%)
- Exhibits20: Historic Market Size - Data Table on Global Influenza Diagnostics Market 2018 - 2022 ($ million)
- Exhibits21: Historic Market Size - End-user Segment 2018 - 2022 ($ million)
- Exhibits22: Historic Market Size - Type Segment 2018 - 2022 ($ million)
- Exhibits23: Historic Market Size - Geography Segment 2018 - 2022 ($ million)
- Exhibits24: Historic Market Size - Country Segment 2018 - 2022 ($ million)
- Exhibits25: Five forces analysis - Comparison between 2023 and 2028
- Exhibits26: Bargaining power of buyers - Impact of key factors 2023 and 2028
- Exhibits27: Bargaining power of suppliers - Impact of key factors in 2023 and 2028
- Exhibits28: Threat of new entrants - Impact of key factors in 2023 and 2028
- Exhibits29: Threat of substitutes - Impact of key factors in 2023 and 2028
- Exhibits30: Threat of rivalry - Impact of key factors in 2023 and 2028
- Exhibits31: Chart on Market condition - Five forces 2023 and 2028
- Exhibits32: Chart on End-user - Market share 2023-2028 (%)
- Exhibits33: Data Table on End-user - Market share 2023-2028 (%)
- Exhibits34: Chart on Comparison by End-user
- Exhibits35: Data Table on Comparison by End-user
- Exhibits36: Chart on Hospitals and diagnostic laboratories - Market size and forecast 2023-2028 ($ million)
- Exhibits37: Data Table on Hospitals and diagnostic laboratories - Market size and forecast 2023-2028 ($ million)
- Exhibits38: Chart on Hospitals and diagnostic laboratories - Year-over-year growth 2023-2028 (%)
- Exhibits39: Data Table on Hospitals and diagnostic laboratories - Year-over-year growth 2023-2028 (%)
- Exhibits40: Chart on Academic and research centers - Market size and forecast 2023-2028 ($ million)
- Exhibits41: Data Table on Academic and research centers - Market size and forecast 2023-2028 ($ million)
- Exhibits42: Chart on Academic and research centers - Year-over-year growth 2023-2028 (%)
- Exhibits43: Data Table on Academic and research centers - Year-over-year growth 2023-2028 (%)
- Exhibits44: Chart on Homecare - Market size and forecast 2023-2028 ($ million)
- Exhibits45: Data Table on Homecare - Market size and forecast 2023-2028 ($ million)
- Exhibits46: Chart on Homecare - Year-over-year growth 2023-2028 (%)
- Exhibits47: Data Table on Homecare - Year-over-year growth 2023-2028 (%)
- Exhibits48: Market opportunity by End-user ($ million)
- Exhibits49: Data Table on Market opportunity by End-user ($ million)
- Exhibits50: Chart on Type - Market share 2023-2028 (%)
- Exhibits51: Data Table on Type - Market share 2023-2028 (%)
- Exhibits52: Chart on Comparison by Type
- Exhibits53: Data Table on Comparison by Type
- Exhibits54: Chart on Point-of-care testing - Market size and forecast 2023-2028 ($ million)
- Exhibits55: Data Table on Point-of-care testing - Market size and forecast 2023-2028 ($ million)
- Exhibits56: Chart on Point-of-care testing - Year-over-year growth 2023-2028 (%)
- Exhibits57: Data Table on Point-of-care testing - Year-over-year growth 2023-2028 (%)
- Exhibits58: Chart on Immunodiagnostics - Market size and forecast 2023-2028 ($ million)
- Exhibits59: Data Table on Immunodiagnostics - Market size and forecast 2023-2028 ($ million)
- Exhibits60: Chart on Immunodiagnostics - Year-over-year growth 2023-2028 (%)
- Exhibits61: Data Table on Immunodiagnostics - Year-over-year growth 2023-2028 (%)
- Exhibits62: Chart on Molecular diagnostics - Market size and forecast 2023-2028 ($ million)
- Exhibits63: Data Table on Molecular diagnostics - Market size and forecast 2023-2028 ($ million)
- Exhibits64: Chart on Molecular diagnostics - Year-over-year growth 2023-2028 (%)
- Exhibits65: Data Table on Molecular diagnostics - Year-over-year growth 2023-2028 (%)
- Exhibits66: Market opportunity by Type ($ million)
- Exhibits67: Data Table on Market opportunity by Type ($ million)
- Exhibits68: Analysis of price sensitivity, lifecycle, customer purchase basket, adoption rates, and purchase criteria
- Exhibits69: Chart on Market share By Geographical Landscape2023-2028 (%)
- Exhibits70: Data Table on Market share By Geographical Landscape2023-2028 (%)
- Exhibits71: Chart on Geographic comparison
- Exhibits72: Data Table on Geographic comparison
- Exhibits73: Chart on North America - Market size and forecast 2023-2028 ($ million)
- Exhibits74: Data Table on North America - Market size and forecast 2023-2028 ($ million)
- Exhibits75: Chart on North America - Year-over-year growth 2023-2028 (%)
- Exhibits76: Data Table on North America - Year-over-year growth 2023-2028 (%)
- Exhibits77: Chart on Europe - Market size and forecast 2023-2028 ($ million)
- Exhibits78: Data Table on Europe - Market size and forecast 2023-2028 ($ million)
- Exhibits79: Chart on Europe - Year-over-year growth 2023-2028 (%)
- Exhibits80: Data Table on Europe - Year-over-year growth 2023-2028 (%)
- Exhibits81: Chart on Asia - Market size and forecast 2023-2028 ($ million)
- Exhibits82: Data Table on Asia - Market size and forecast 2023-2028 ($ million)
- Exhibits83: Chart on Asia - Year-over-year growth 2023-2028 (%)
- Exhibits84: Data Table on Asia - Year-over-year growth 2023-2028 (%)
- Exhibits85: Chart on Rest of World (ROW) - Market size and forecast 2023-2028 ($ million)
- Exhibits86: Data Table on Rest of World (ROW) - Market size and forecast 2023-2028 ($ million)
- Exhibits87: Chart on Rest of World (ROW) - Year-over-year growth 2023-2028 (%)
- Exhibits88: Data Table on Rest of World (ROW) - Year-over-year growth 2023-2028 (%)
- Exhibits89: Chart on US - Market size and forecast 2023-2028 ($ million)
- Exhibits90: Data Table on US - Market size and forecast 2023-2028 ($ million)
- Exhibits91: Chart on US - Year-over-year growth 2023-2028 (%)
- Exhibits92: Data Table on US - Year-over-year growth 2023-2028 (%)
- Exhibits93: Chart on China - Market size and forecast 2023-2028 ($ million)
- Exhibits94: Data Table on China - Market size and forecast 2023-2028 ($ million)
- Exhibits95: Chart on China - Year-over-year growth 2023-2028 (%)
- Exhibits96: Data Table on China - Year-over-year growth 2023-2028 (%)
- Exhibits97: Chart on Germany - Market size and forecast 2023-2028 ($ million)
- Exhibits98: Data Table on Germany - Market size and forecast 2023-2028 ($ million)
- Exhibits99: Chart on Germany - Year-over-year growth 2023-2028 (%)
- Exhibits100: Data Table on Germany - Year-over-year growth 2023-2028 (%)
- Exhibits101: Chart on France - Market size and forecast 2023-2028 ($ million)
- Exhibits102: Data Table on France - Market size and forecast 2023-2028 ($ million)
- Exhibits103: Chart on France - Year-over-year growth 2023-2028 (%)
- Exhibits104: Data Table on France - Year-over-year growth 2023-2028 (%)
- Exhibits105: Chart on Canada - Market size and forecast 2023-2028 ($ million)
- Exhibits106: Data Table on Canada - Market size and forecast 2023-2028 ($ million)
- Exhibits107: Chart on Canada - Year-over-year growth 2023-2028 (%)
- Exhibits108: Data Table on Canada - Year-over-year growth 2023-2028 (%)
- Exhibits109: Market opportunity By Geographical Landscape($ million)
- Exhibits110: Data Tables on Market opportunity By Geographical Landscape($ million)
- Exhibits111: Impact of drivers and challenges in 2023 and 2028
- Exhibits112: Overview on criticality of inputs and factors of differentiation
- Exhibits113: Overview on factors of disruption
- Exhibits114: Impact of key risks on business
- Exhibits115: Companies covered
- Exhibits116: Matrix on companies position and classification
- Exhibits117: Abbott Laboratories - Overview
- Exhibits118: Abbott Laboratories - Business segments
- Exhibits119: Abbott Laboratories - Key news
- Exhibits120: Abbott Laboratories - Key offerings
- Exhibits121: Abbott Laboratories - Segment focus
- Exhibits122: Becton Dickinson and Co. - Overview
- Exhibits123: Becton Dickinson and Co. - Business segments
- Exhibits124: Becton Dickinson and Co. - Key news
- Exhibits125: Becton Dickinson and Co. - Key offerings
- Exhibits126: Becton Dickinson and Co. - Segment focus
- Exhibits127: BICO Group AB - Overview
- Exhibits128: BICO Group AB - Business segments
- Exhibits129: BICO Group AB - Key news
- Exhibits130: BICO Group AB - Key offerings
- Exhibits131: BICO Group AB - Segment focus
- Exhibits132: Bio Rad Laboratories Inc. - Overview
- Exhibits133: Bio Rad Laboratories Inc. - Business segments
- Exhibits134: Bio Rad Laboratories Inc. - Key news
- Exhibits135: Bio Rad Laboratories Inc. - Key offerings
- Exhibits136: Bio Rad Laboratories Inc. - Segment focus
- Exhibits137: Biocartis NV - Overview
- Exhibits138: Biocartis NV - Product / Service
- Exhibits139: Biocartis NV - Key news
- Exhibits140: Biocartis NV - Key offerings
- Exhibits141: bioMerieux SA - Overview
- Exhibits142: bioMerieux SA - Product / Service
- Exhibits143: bioMerieux SA - Key news
- Exhibits144: bioMerieux SA - Key offerings
- Exhibits145: Danaher Corp. - Overview
- Exhibits146: Danaher Corp. - Business segments
- Exhibits147: Danaher Corp. - Key news
- Exhibits148: Danaher Corp. - Key offerings
- Exhibits149: Danaher Corp. - Segment focus
- Exhibits150: DiaSorin SpA - Overview
- Exhibits151: DiaSorin SpA - Business segments
- Exhibits152: DiaSorin SpA - Key news
- Exhibits153: DiaSorin SpA - Key offerings
- Exhibits154: DiaSorin SpA - Segment focus
- Exhibits155: F. Hoffmann La Roche Ltd. - Overview
- Exhibits156: F. Hoffmann La Roche Ltd. - Business segments
- Exhibits157: F. Hoffmann La Roche Ltd. - Key news
- Exhibits158: F. Hoffmann La Roche Ltd. - Key offerings
- Exhibits159: F. Hoffmann La Roche Ltd. - Segment focus
- Exhibits160: Hologic Inc. - Overview
- Exhibits161: Hologic Inc. - Business segments
- Exhibits162: Hologic Inc. - Key news
- Exhibits163: Hologic Inc. - Key offerings
- Exhibits164: Hologic Inc. - Segment focus
- Exhibits165: Princeton BioMeditech Corp. - Overview
- Exhibits166: Princeton BioMeditech Corp. - Product / Service
- Exhibits167: Princeton BioMeditech Corp. - Key offerings
- Exhibits168: Quidelortho Corp. - Overview
- Exhibits169: Quidelortho Corp. - Product / Service
- Exhibits170: Quidelortho Corp. - Key news
- Exhibits171: Quidelortho Corp. - Key offerings
- Exhibits172: Sd Biosensor Inc. - Overview
- Exhibits173: Sd Biosensor Inc. - Product / Service
- Exhibits174: Sd Biosensor Inc. - Key offerings
- Exhibits175: Siemens Healthineers AG - Overview
- Exhibits176: Siemens Healthineers AG - Business segments
- Exhibits177: Siemens Healthineers AG - Key news
- Exhibits178: Siemens Healthineers AG - Key offerings
- Exhibits179: Siemens Healthineers AG - Segment focus
- Exhibits180: Thermo Fisher Scientific Inc. - Overview
- Exhibits181: Thermo Fisher Scientific Inc. - Business segments
- Exhibits182: Thermo Fisher Scientific Inc. - Key news
- Exhibits183: Thermo Fisher Scientific Inc. - Key offerings
- Exhibits184: Thermo Fisher Scientific Inc. - Segment focus
- Exhibits185: Inclusions checklist
- Exhibits186: Exclusions checklist
- Exhibits187: Currency conversion rates for US$
- Exhibits188: Research methodology
- Exhibits189: Information sources
- Exhibits190: Data validation
- Exhibits191: Validation techniques employed for market sizing
- Exhibits192: Data synthesis
- Exhibits193: 360 degree market analysis
- Exhibits194: List of abbreviations
The influenza diagnostics market is forecasted to grow by USD 517.5 mn during 2023-2028, accelerating at a CAGR of 7.22% during the forecast period. The report on the influenza diagnostics market provides a holistic analysis, market size and forecast, trends, growth drivers, and challenges, as well as vendor analysis covering around 25 vendors.
The report offers an up-to-date analysis regarding the current market scenario, the latest trends and drivers, and the overall market environment. The market is driven by rising incidence of influenza, growing demand for molecular diagnostics, and supportive government initiatives and funding.
Technavio's influenza diagnostics market is segmented as below:
| Market Scope | |
|---|---|
| Base Year | 2024 |
| End Year | 2028 |
| Series Year | 2024-2028 |
| Growth Momentum | Accelerate |
| YOY 2024 | 6.6% |
| CAGR | 7.22% |
| Incremental Value | $517.5mn |
By End-user
- Hospitals and diagnostic laboratories
- Academic and research centers
- Homecare
By Type
- Point-of-care testing
- Immunodiagnostics
- Molecular diagnostics
By Geography
- North America
- Europe
- Asia
- Rest of World (ROW)
This study identifies the rising approvals for influenza diagnostic products as one of the prime reasons driving the influenza diagnostics market growth during the next few years. Also, surge in number of genetic characterization studies on influenza virus and rising adoption of rapid diagnostic products in physician office laboratories will lead to sizable demand in the market.
The report on the influenza diagnostics market covers the following areas:
- Influenza diagnostics market sizing
- Influenza diagnostics market forecast
- Influenza diagnostics market industry analysis
The robust vendor analysis is designed to help clients improve their market position, and in line with this, this report provides a detailed analysis of several leading influenza diagnostics market vendors that include Abbott Laboratories, Becton Dickinson and Co., BICO Group AB, Bio Rad Laboratories Inc., Biocartis NV, bioMerieux SA, Danaher Corp., DiaSorin SpA, F. Hoffmann La Roche Ltd., Hologic Inc., Princeton BioMeditech Corp., QIAGEN NV, Quidelortho Corp., Response Biomedical Corp., SA Scientific Ltd., Sd Biosensor Inc., Sekisui Chemical Co. Ltd., Siemens Healthineers AG, Tecan Trading AG, and Thermo Fisher Scientific Inc.. Also, the influenza diagnostics market analysis report includes information on upcoming trends and challenges that will influence market growth. This is to help companies strategize and leverage all forthcoming growth opportunities.
The study was conducted using an objective combination of primary and secondary information including inputs from key participants in the industry. The report contains a comprehensive market and vendor landscape in addition to an analysis of the key vendors.
The publisher presents a detailed picture of the market by the way of study, synthesis, and summation of data from multiple sources by an analysis of key parameters such as profit, pricing, competition, and promotions. It presents various market facets by identifying the key industry influencers. The data presented is comprehensive, reliable, and a result of extensive research - both primary and secondary. The market research reports provide a complete competitive landscape and an in-depth vendor selection methodology and analysis using qualitative and quantitative research to forecast the accurate market growth.
Table of Contents
1 Executive Summary
- 1.1 Market overview
- Executive Summary - Chart on Market Overview
- Executive Summary - Data Table on Market Overview
- Executive Summary - Chart on Global Market Characteristics
- Executive Summary - Chart on Market by Geography
- Executive Summary - Chart on Market Segmentation by End-user
- Executive Summary - Chart on Market Segmentation by Type
- Executive Summary - Chart on Incremental Growth
- Executive Summary - Data Table on Incremental Growth
- Executive Summary - Chart on Company Market Positioning
2 Market Landscape
- 2.1 Market ecosystem
- Parent Market
- Data Table on - Parent Market
- 2.2 Market characteristics
- Market characteristics analysis
- 2.3 Value chain analysis
- Value Chain Analysis
3 Market Sizing
- 3.1 Market definition
- Offerings of companies included in the market definition
- 3.2 Market segment analysis
- Market segments
- 3.3 Market size 2023
- 3.4 Market outlook: Forecast for 2023-2028
- Chart on Global - Market size and forecast 2023-2028 ($ million)
- Data Table on Global - Market size and forecast 2023-2028 ($ million)
- Chart on Global Market: Year-over-year growth 2023-2028 (%)
- Data Table on Global Market: Year-over-year growth 2023-2028 (%)
4 Historic Market Size
- 4.1 Global Influenza Diagnostics Market 2018 - 2022
- Historic Market Size - Data Table on Global Influenza Diagnostics Market 2018 - 2022 ($ million)
- 4.2 End-user segment analysis 2018 - 2022
- Historic Market Size - End-user Segment 2018 - 2022 ($ million)
- 4.3 Type segment analysis 2018 - 2022
- Historic Market Size - Type Segment 2018 - 2022 ($ million)
- 4.4 Geography segment analysis 2018 - 2022
- Historic Market Size - Geography Segment 2018 - 2022 ($ million)
- 4.5 Country segment analysis 2018 - 2022
- Historic Market Size - Country Segment 2018 - 2022 ($ million)
5 Five Forces Analysis
- 5.1 Five forces summary
- Five forces analysis - Comparison between 2023 and 2028
- 5.2 Bargaining power of buyers
- Bargaining power of buyers - Impact of key factors 2023 and 2028
- 5.3 Bargaining power of suppliers
- Bargaining power of suppliers - Impact of key factors in 2023 and 2028
- 5.4 Threat of new entrants
- Threat of new entrants - Impact of key factors in 2023 and 2028
- 5.5 Threat of substitutes
- Threat of substitutes - Impact of key factors in 2023 and 2028
- 5.6 Threat of rivalry
- Threat of rivalry - Impact of key factors in 2023 and 2028
- 5.7 Market condition
- Chart on Market condition - Five forces 2023 and 2028
6 Market Segmentation by End-user
- 6.1 Market segments
- Chart on End-user - Market share 2023-2028 (%)
- Data Table on End-user - Market share 2023-2028 (%)
- 6.2 Comparison by End-user
- Chart on Comparison by End-user
- Data Table on Comparison by End-user
- 6.3 Hospitals and diagnostic laboratories - Market size and forecast 2023-2028
- Chart on Hospitals and diagnostic laboratories - Market size and forecast 2023-2028 ($ million)
- Data Table on Hospitals and diagnostic laboratories - Market size and forecast 2023-2028 ($ million)
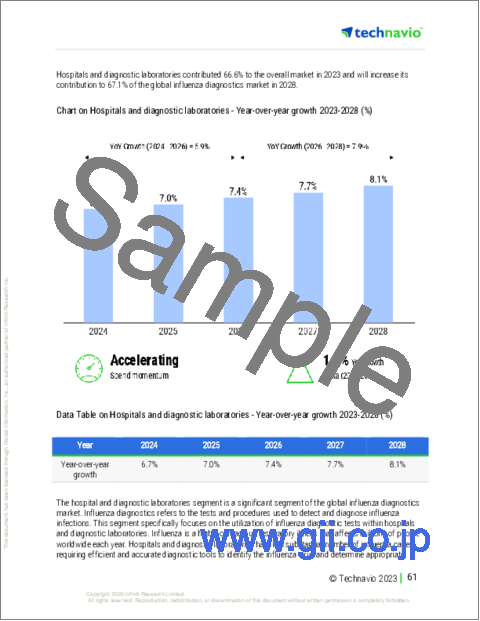
- Chart on Hospitals and diagnostic laboratories - Year-over-year growth 2023-2028 (%)
- Data Table on Hospitals and diagnostic laboratories - Year-over-year growth 2023-2028 (%)
- 6.4 Academic and research centers - Market size and forecast 2023-2028
- Chart on Academic and research centers - Market size and forecast 2023-2028 ($ million)
- Data Table on Academic and research centers - Market size and forecast 2023-2028 ($ million)
- Chart on Academic and research centers - Year-over-year growth 2023-2028 (%)
- Data Table on Academic and research centers - Year-over-year growth 2023-2028 (%)
- 6.5 Homecare - Market size and forecast 2023-2028
- Chart on Homecare - Market size and forecast 2023-2028 ($ million)
- Data Table on Homecare - Market size and forecast 2023-2028 ($ million)
- Chart on Homecare - Year-over-year growth 2023-2028 (%)
- Data Table on Homecare - Year-over-year growth 2023-2028 (%)
- 6.6 Market opportunity by End-user
- Market opportunity by End-user ($ million)
- Data Table on Market opportunity by End-user ($ million)
7 Market Segmentation by Type
- 7.1 Market segments
- Chart on Type - Market share 2023-2028 (%)
- Data Table on Type - Market share 2023-2028 (%)
- 7.2 Comparison by Type
- Chart on Comparison by Type
- Data Table on Comparison by Type
- 7.3 Point-of-care testing - Market size and forecast 2023-2028
- Chart on Point-of-care testing - Market size and forecast 2023-2028 ($ million)
- Data Table on Point-of-care testing - Market size and forecast 2023-2028 ($ million)
- Chart on Point-of-care testing - Year-over-year growth 2023-2028 (%)
- Data Table on Point-of-care testing - Year-over-year growth 2023-2028 (%)
- 7.4 Immunodiagnostics - Market size and forecast 2023-2028
- Chart on Immunodiagnostics - Market size and forecast 2023-2028 ($ million)
- Data Table on Immunodiagnostics - Market size and forecast 2023-2028 ($ million)
- Chart on Immunodiagnostics - Year-over-year growth 2023-2028 (%)
- Data Table on Immunodiagnostics - Year-over-year growth 2023-2028 (%)
- 7.5 Molecular diagnostics - Market size and forecast 2023-2028
- Chart on Molecular diagnostics - Market size and forecast 2023-2028 ($ million)
- Data Table on Molecular diagnostics - Market size and forecast 2023-2028 ($ million)
- Chart on Molecular diagnostics - Year-over-year growth 2023-2028 (%)
- Data Table on Molecular diagnostics - Year-over-year growth 2023-2028 (%)
- 7.6 Market opportunity by Type
- Market opportunity by Type ($ million)
- Data Table on Market opportunity by Type ($ million)
8 Customer Landscape
- 8.1 Customer landscape overview
- Analysis of price sensitivity, lifecycle, customer purchase basket, adoption rates, and purchase criteria
9 Geographic Landscape
- 9.1 Geographic segmentation
- Chart on Market share By Geographical Landscape2023-2028 (%)
- Data Table on Market share By Geographical Landscape2023-2028 (%)
- 9.2 Geographic comparison
- Chart on Geographic comparison
- Data Table on Geographic comparison
- 9.3 North America - Market size and forecast 2023-2028
- Chart on North America - Market size and forecast 2023-2028 ($ million)
- Data Table on North America - Market size and forecast 2023-2028 ($ million)
- Chart on North America - Year-over-year growth 2023-2028 (%)
- Data Table on North America - Year-over-year growth 2023-2028 (%)
- 9.4 Europe - Market size and forecast 2023-2028
- Chart on Europe - Market size and forecast 2023-2028 ($ million)
- Data Table on Europe - Market size and forecast 2023-2028 ($ million)
- Chart on Europe - Year-over-year growth 2023-2028 (%)
- Data Table on Europe - Year-over-year growth 2023-2028 (%)
- 9.5 Asia - Market size and forecast 2023-2028
- Chart on Asia - Market size and forecast 2023-2028 ($ million)
- Data Table on Asia - Market size and forecast 2023-2028 ($ million)
- Chart on Asia - Year-over-year growth 2023-2028 (%)
- Data Table on Asia - Year-over-year growth 2023-2028 (%)
- 9.6 Rest of World (ROW) - Market size and forecast 2023-2028
- Chart on Rest of World (ROW) - Market size and forecast 2023-2028 ($ million)
- Data Table on Rest of World (ROW) - Market size and forecast 2023-2028 ($ million)
- Chart on Rest of World (ROW) - Year-over-year growth 2023-2028 (%)
- Data Table on Rest of World (ROW) - Year-over-year growth 2023-2028 (%)
- 9.7 US - Market size and forecast 2023-2028
- Chart on US - Market size and forecast 2023-2028 ($ million)
- Data Table on US - Market size and forecast 2023-2028 ($ million)
- Chart on US - Year-over-year growth 2023-2028 (%)
- Data Table on US - Year-over-year growth 2023-2028 (%)
- 9.8 China - Market size and forecast 2023-2028
- Chart on China - Market size and forecast 2023-2028 ($ million)
- Data Table on China - Market size and forecast 2023-2028 ($ million)
- Chart on China - Year-over-year growth 2023-2028 (%)
- Data Table on China - Year-over-year growth 2023-2028 (%)
- 9.9 Germany - Market size and forecast 2023-2028
- Chart on Germany - Market size and forecast 2023-2028 ($ million)
- Data Table on Germany - Market size and forecast 2023-2028 ($ million)
- Chart on Germany - Year-over-year growth 2023-2028 (%)
- Data Table on Germany - Year-over-year growth 2023-2028 (%)
- 9.10 France - Market size and forecast 2023-2028
- Chart on France - Market size and forecast 2023-2028 ($ million)
- Data Table on France - Market size and forecast 2023-2028 ($ million)
- Chart on France - Year-over-year growth 2023-2028 (%)
- Data Table on France - Year-over-year growth 2023-2028 (%)
- 9.11 Canada - Market size and forecast 2023-2028
- Chart on Canada - Market size and forecast 2023-2028 ($ million)
- Data Table on Canada - Market size and forecast 2023-2028 ($ million)
- Chart on Canada - Year-over-year growth 2023-2028 (%)
- Data Table on Canada - Year-over-year growth 2023-2028 (%)
- 9.12 Market opportunity by geography
- Market opportunity By Geographical Landscape($ million)
- Data Tables on Market opportunity By Geographical Landscape($ million)
10 Drivers, Challenges, and Opportunity/Restraints
- 10.1 Market drivers
- 10.2 Market challenges
- 10.3 Impact of drivers and challenges
- Impact of drivers and challenges in 2023 and 2028
- 10.4 Market opportunities/restraints
11 Competitive Landscape
- 11.1 Overview
- 11.2 Competitive Landscape
- Overview on criticality of inputs and factors of differentiation
- 11.3 Landscape disruption
- Overview on factors of disruption
- 11.4 Industry risks
- Impact of key risks on business
12 Competitive Analysis
- 12.1 Companies profiled
- Companies covered
- 12.2 Market positioning of companies
- Matrix on companies position and classification
- 12.3 Abbott Laboratories
- Abbott Laboratories - Overview
- Abbott Laboratories - Business segments
- Abbott Laboratories - Key news
- Abbott Laboratories - Key offerings
- Abbott Laboratories - Segment focus
- 12.4 Becton Dickinson and Co.
- Becton Dickinson and Co. - Overview
- Becton Dickinson and Co. - Business segments
- Becton Dickinson and Co. - Key news
- Becton Dickinson and Co. - Key offerings
- Becton Dickinson and Co. - Segment focus
- 12.5 BICO Group AB
- BICO Group AB - Overview
- BICO Group AB - Business segments
- BICO Group AB - Key news
- BICO Group AB - Key offerings
- BICO Group AB - Segment focus
- 12.6 Bio Rad Laboratories Inc.
- Bio Rad Laboratories Inc. - Overview
- Bio Rad Laboratories Inc. - Business segments
- Bio Rad Laboratories Inc. - Key news
- Bio Rad Laboratories Inc. - Key offerings
- Bio Rad Laboratories Inc. - Segment focus
- 12.7 Biocartis NV
- Biocartis NV - Overview
- Biocartis NV - Product / Service
- Biocartis NV - Key news
- Biocartis NV - Key offerings
- 12.8 bioMerieux SA
- bioMerieux SA - Overview
- bioMerieux SA - Product / Service
- bioMerieux SA - Key news
- bioMerieux SA - Key offerings
- 12.9 Danaher Corp.
- Danaher Corp. - Overview
- Danaher Corp. - Business segments
- Danaher Corp. - Key news
- Danaher Corp. - Key offerings
- Danaher Corp. - Segment focus
- 12.10 DiaSorin SpA
- DiaSorin SpA - Overview
- DiaSorin SpA - Business segments
- DiaSorin SpA - Key news
- DiaSorin SpA - Key offerings
- DiaSorin SpA - Segment focus
- 12.11 F. Hoffmann La Roche Ltd.
- F. Hoffmann La Roche Ltd. - Overview
- F. Hoffmann La Roche Ltd. - Business segments
- F. Hoffmann La Roche Ltd. - Key news
- F. Hoffmann La Roche Ltd. - Key offerings
- F. Hoffmann La Roche Ltd. - Segment focus
- 12.12 Hologic Inc.
- Hologic Inc. - Overview
- Hologic Inc. - Business segments
- Hologic Inc. - Key news
- Hologic Inc. - Key offerings
- Hologic Inc. - Segment focus
- 12.13 Princeton BioMeditech Corp.
- Princeton BioMeditech Corp. - Overview
- Princeton BioMeditech Corp. - Product / Service
- Princeton BioMeditech Corp. - Key offerings
- 12.14 Quidelortho Corp.
- Quidelortho Corp. - Overview
- Quidelortho Corp. - Product / Service
- Quidelortho Corp. - Key news
- Quidelortho Corp. - Key offerings
- 12.15 Sd Biosensor Inc.
- Sd Biosensor Inc. - Overview
- Sd Biosensor Inc. - Product / Service
- Sd Biosensor Inc. - Key offerings
- 12.16 Siemens Healthineers AG
- Siemens Healthineers AG - Overview
- Siemens Healthineers AG - Business segments
- Siemens Healthineers AG - Key news
- Siemens Healthineers AG - Key offerings
- Siemens Healthineers AG - Segment focus
- 12.17 Thermo Fisher Scientific Inc.
- Thermo Fisher Scientific Inc. - Overview
- Thermo Fisher Scientific Inc. - Business segments
- Thermo Fisher Scientific Inc. - Key news
- Thermo Fisher Scientific Inc. - Key offerings
- Thermo Fisher Scientific Inc. - Segment focus
13 Appendix
- 13.1 Scope of the report
- 13.2 Inclusions and exclusions checklist
- Inclusions checklist
- Exclusions checklist
- 13.3 Currency conversion rates for US$
- Currency conversion rates for US$
- 13.4 Research methodology
- Research methodology
- 13.5 Data procurement
- Information sources
- 13.6 Data validation
- Data validation
- 13.7 Validation techniques employed for market sizing
- Validation techniques employed for market sizing
- 13.8 Data synthesis
- Data synthesis
- 13.9 360 degree market analysis
- 360 degree market analysis
- 13.10 List of abbreviations
- List of abbreviations