|
|
市場調査レポート
商品コード
1164879
食道がんの世界市場:上市済み薬品とパイプライン薬品の評価、臨床試験、競合情勢Esophageal Cancer Marketed and Pipeline Drugs Assessment, Clinical Trials and Competitive Landscape |
||||||
|
● お客様のご希望に応じて、既存データの加工や未掲載情報(例:国別セグメント)の追加などの対応が可能です。 詳細はお問い合わせください。 |
|||||||
価格
| 食道がんの世界市場:上市済み薬品とパイプライン薬品の評価、臨床試験、競合情勢 |
|
出版日: 2022年10月28日
発行: GlobalData
ページ情報: 英文 73 Pages
納期: 即納可能
|
ご注意事項 :
本レポートは最新情報反映のため適宜更新し、内容構成変更を行う場合があります。ご検討の際はお問い合わせください。
- 全表示
- 概要
- 目次
概要
当レポートでは、世界の食道がん市場について調査分析し、疾患の情勢、上市済み薬品とパイプライン薬品の評価、現在と将来の競合情勢などを提供しています。
目次
目次
第1章 序文
第2章 主な調査結果
第3章 疾患の情勢
- 疾患の概要
- 疫学の概要
- 治療の概要
第4章 上市済み薬品の評価
- 主な上市済み薬品
- 概要:作用機序別
- 概要:投与経路別
- 上市済み薬品のプロファイルと販売予測
第5章 価格設定と償還の評価
- メーカー価格
- 価格設定と償還までの時間
第6章 パイプライン薬品の評価
- 第III相パイプライン薬品
- 概要:開発段階別
- 概要:分子タイプ別
- 概要:作用機序別
- 概要:投与経路別
- 薬品固有の相移行成功率(PTSR)・承認の可能性(LoA)
- 治療領域と適応症固有のPTSR・LoA
第7章 臨床試験の評価
- 過去の概要
- 概要:段階別
- 概要:ステータス別
- 概要:進行中・計画中の試験の段階別
- 仮想コンポーネントを使用した試験
- 地理的概要
- 単一国・多国間試験:地域別
- 上位20のスポンサー:段階別
- 上位20のスポンサー:ステータス別
- 概要:エンドポイントステータス別
- 概要:人種・民族別
- 登録データ
- 試験施設の上位20か国
- 世界の上位20施設
- 実現可能性分析 - 地理的概要
- 実現可能性分析 - ベンチマークモデル
第8章 取引情勢
- 合併、買収、戦略的提携:地域別
- 最近の合併、買収、戦略的提携
第9章 商業的評価
- 主な市場企業
第10章 将来の市場カタリスト
第11章 付録
目次
Product Code: GDHC086CL
This reports provides a data-driven overview of the current and future competitive landscape in Esophageal Cancer therapeutics.
Synopsis
- In 2022, there will be more than 440,000 diagnosed prevalent cases of esophageal cancer across 16 pharmaceutical markets.
- Currently there are four leading marketed innovator drugs for the treatment of esophageal cancer, mostly programmed cell death protein 1 (PD1) antagonists..
- A total of 14 pipeline drugs for esophageal cancer are in Phase III and two drugs are in pre-registration stage of development.
- Non-commercial sponsors dominate clinical trial development in esophageal cancer, with China emerging as the key country for conducting Phase III trials in this disease space.
- In deals involving companies developing esophageal cancer assets, there were 18 completed or announced strategic alliances and four merger and acquisition transactions.
- Emerging players in the esophageal cancer treatment space are focused on launching novel investigational drugs for treatment on a global scale.
Scope
GlobalData's Esophageal Cancer Marketed and Pipeline Drugs Assessment, Clinical Trials and Competitive Landscape combines data from the Pharma Intelligence Center with in-house analyst expertise to provide a competitive assessment of the disease marketplace.
Components of the report include -
- Disease Landscape
- Disease Overview
- Epidemiology Overview
- Treatment Overview
- Marketed Products Assessment
- Breakdown by Mechanism of Action, Molecule Type, Route of Administration
- Product Profiles with Sales Forecast
- Pricing and Reimbursement Assessment
- Retail or Manufacturer Price of Products
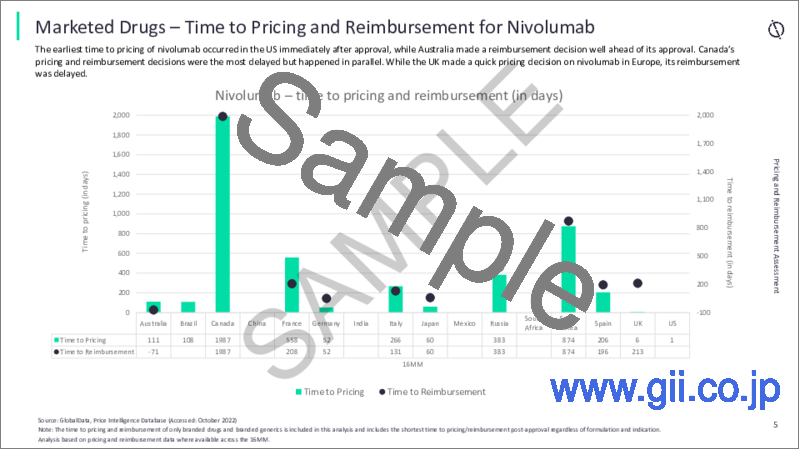
- Time to Pricing and Time to Reimbursement
- Pipeline Assessment
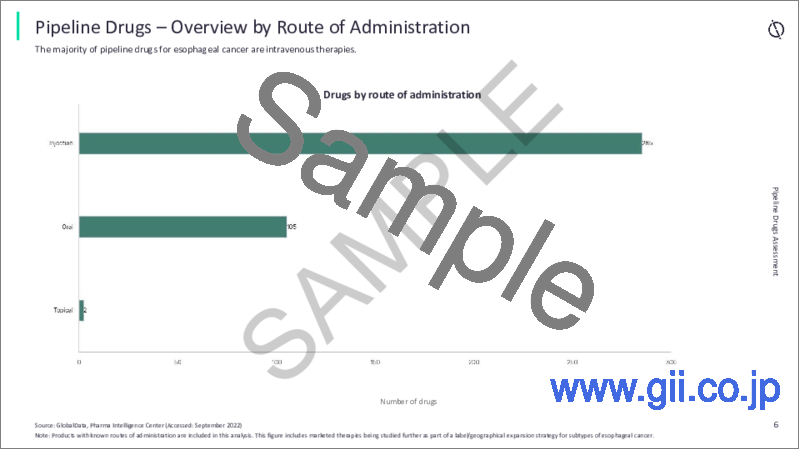
- Breakdown by Development Stage, Mechanism of Action, Molecule Type, Route of Administration
- Product Profiles with Sales Forecast
- Late-to-mid-stage Pipeline Drugs
- Phase Transition Success Rate and Likelihood of Approval
- Clinical Trials Assessment
- Breakdown of Trials by Phase, Status, Virtual Components, Sponsors, Geography, and Endpoint Status
- Enrolment Analytics, Site Analytics, Feasibility Analysis
- Deals Landscape
- Mergers, Acquisitions, and Strategic Alliances by Region
- Overview of Recent Deals
- Commercial Assessment
- Key Market Players
- Future Market Catalysts
Reasons to Buy
- Develop and design your in-licensing and out-licensing strategies through a review of pipeline products and technologies, and by identifying the companies with the most robust pipeline.
- Develop business strategies by understanding the trends shaping and driving the Esophageal Cancer market.
- Drive revenues by understanding the key trends, innovative products and technologies, market segments, and companies likely to impact the global Esophageal Cancer market in the future.
- Formulate effective sales and marketing strategies by understanding the competitive landscape and by analyzing the performance of various competitors.
- Identify emerging players with potentially strong product portfolios and create effective counter-strategies to gain a competitive advantage.
- Organize your sales and marketing efforts by identifying the market categories and segments that present maximum opportunities for consolidations, investments, and strategic partnerships.
Table of Contents
Table of Contents
1 Preface
- 1.1 Contents
- 1.2 Report Scope
- 1.3 List of Tables and Figures
- 1.4 Abbreviations
2 Key Findings
3 Disease Landscape
- 3.1 Disease Overview
- 3.2 Epidemiology Overview
- 3.3 Treatment Overview
4 Marketed Drugs Assessment
- 4.1 Leading Marketed Drugs
- 4.2 Overview by Mechanism of Action
- 4.3 Overview by Route of Administration
- 4.4 Marketed Drugs Profiles and Sales Forecasts
5 Pricing and Reimbursement Assessment
- 5.1 Manufacturer Price
- 5.2 Time to Pricing and Reimbursement
6 Pipeline Drugs Assessment
- 6.1 Phase III Pipeline Drugs
- 6.2 Overview by Development Stage
- 6.3 Overview by Molecule Type
- 6.4 Overview by Mechanism of Action
- 6.5 Overview by Route of Administration
- 6.6 Drug Specific Phase Transition Success Rate (PTSR) and Likelihood of Approval (LoA)
- 6.7 Therapy Area and Indication-specific PTSR and LoA
7 Clinical Trials Assessment
- 7.1 Historical Overview
- 7.2 Overview by Phase
- 7.3 Overview by Status
- 7.4 Overview by Phase for Ongoing and Planned Trials
- 7.5 Trials with Virtual Components
- 7.6 Geographic Overview
- 7.7 Single-Country and Multinational Trials by Region
- 7.8 Top 20 Sponsors with Breakdown by Phase
- 7.9 Top 20 Sponsors with Breakdown by Status
- 7.10 Overview by Endpoint Status
- 7.11 Overview by Race and Ethnicity
- 7.12 Enrollment Data
- 7.13 Top 20 countries for Trial Sites
- 7.14 Top 20 Sites Globally
- 7.15 Feasibility Analysis - Geographic Overview
- 7.16 Feasibility Analysis - Benchmark Models
8 Deals Landscape
- 8.1 Mergers, Acquisitions, and Strategic Alliances by Region
- 8.2 Recent Mergers, Acquisitions, and Strategic Alliances
9 Commercial Assessment
- 9.1 Key Market Players
10 Future Market Catalysts
11 Appendix
- 11.1 Methodology
- 11.2 Methodology - Sales Forecast
- 11.3 Methodology - Pricing and Reimbursement
- 11.4 Methodology - PTSR and LoA Analysis
- 11.5 About the Authors
- 11.6 Contact Us
- 11.7 Disclaimer
お電話でのお問い合わせ
044-952-0102
( 土日・祝日を除く )