|
|
市場調査レポート
商品コード
1286685
フェニルケトン尿症市場 - 市場の洞察、疫学、市場予測:2032年Phenylketonuria Market Insight, Epidemiology And Market Forecast - 2032 |
||||||
カスタマイズ可能
適宜更新あり
|
|||||||
| フェニルケトン尿症市場 - 市場の洞察、疫学、市場予測:2032年 |
|
出版日: 2023年06月01日
発行: DelveInsight
ページ情報: 英文 128 Pages
納期: 2~10営業日
|
- 全表示
- 概要
- 図表
- 目次
2022年、フェニルケトン尿症(PKU)の市場規模は主要7ヶ国で6億1,000万米ドルとなりました。同市場は今後、予測期間中(2023年~2032年)に拡大すると予測されています。
当レポートでは、主要7ヶ国におけるフェニルケトン尿症市場について調査し、市場の概要とともに、疫学、患者動向、新たな治療法、2032年までの市場規模予測、および医療のアンメットニーズなどを提供しています。
目次
第1章 重要な洞察
第2章 レポートのイントロダクション
第3章 フェニルケトン尿症(PKU)市場概要
- 市場シェア(%)2019年のフェニルケトン尿症(PKU)の分布
- 市場シェア(%)2032年のフェニルケトン尿症(PKU)の分布
第4章 フェニルケトン尿症(PKU)のエグゼクティブサマリー
第5章 主要な出来事
第6章 疾患の背景と概要
第7章 調査手法
第8章 疫学と患者数
- 主な調査結果
- 仮定と根拠
- 疫学シナリオ:主要7ヶ国
- 米国
- 欧州4ヶ国と英国
- 日本
第9章 患者動向
第10章 上市済み薬剤
第11章 新興薬剤
第12章 フェニルケトン尿症(PKU):主要7ヶ国分析
- 主な調査結果
- 主要な市場予測の前提条件
- 属性分析
- 主要7ヶ国市場の見通し
- 主要7ヶ国規模:PKU
- 米国におけるフェニルケトン尿症の市場規模
- 欧州4ヶ国および英国におけるフェニルケトン尿症の市場規模
- 日本のフェニルケトン尿症の市場規模
第13章 KOLのビュー
第14章 SWOT分析
第15章 アンメットニーズ
第16章 市場アクセスと償還
- 米国
- 欧州4ヶ国と英国
- 日本
第17章 付録
第18章 DelveInsightのサービス内容
第19章 免責事項
第20章 DelveInsightについて
List of Tables
- Table 1: Summary of PKU, Market and Epidemiology (2019-2032)
- Table 2: Key Events
- Table 3: Approved Drugs
- Table 4: Key Recommendations for Patients With Phenylketonuria (PKU)
- Table 5: Key Points and Recommendations to Effectively Manage PKU
- Table 6: Medical and Dietary Guidelines for the Treatment of PKU
- Table 7: Total Diagnosed Prevalent Cases of PKU in the 7MM (2019-2032)
- Table 8: Total Diagnosed Prevalent Cases of PKU in the United States (2019-2032)
- Table 9: Mutation type-specific Cases of PKU in the United States (2019-2032)
- Table 10: Age-specific Cases of PKU in the United States (2019-2032)
- Table 11: Severity-specific Cases of PKU in the United States (2019-2032)
- Table 12: Total Diagnosed Prevalent Cases of PKU in EU4 and the UK (2019-2032)
- Table 13: Mutation type-specific Cases of PKU in EU4 and the UK (2019-2032)
- Table 14: Age-specific Cases of PKU in EU4 and the UK (2019-2032)
- Table 15: Severity-specific Cases of PKU in EU4 and the UK (2019-2032)
- Table 16: Total Diagnosed Prevalent Cases of PKU in Japan (2019-2032)
- Table 17: Mutation type-specific Cases of PKU in Japan (2019-2032)
- Table 18: Age-specific Cases of PKU in Japan (2019-2032)
- Table 19: Severity-specific Cases of PKU in Japan (2019-2032)
- Table 20: Comparison of Marketed Drugs
- Table 21: KUVAN, Clinical Trial Description, 2023
- Table 22: Blood Phe Results in Study 2
- Table 23: Blood Phe Results From Forced Dose-titration in Study 3
- Table 24: PALYNZIQ, Clinical Trial Description, 2023
- Table 25: Comparison of Emerging Drugs Under Development
- Table 26: PTC923, Clinical Trial Description, 2023
- Table 27: SYNB1934, Clinical Trial Description, 2023
- Table 28: Key Market Forecast Assumptions for PTC923
- Table 29: Key Market Forecast Assumptions for SYNB1934
- Table 30: Seven Major Market Size of PKU in USD million (2019-2032)
- Table 31: Seven Major Market Size of PKU by Therapies in USD million (2019-2032)
- Table 32: Total market size of PKU in the United States in USD million (2019-2032)
- Table 33: Market size of PKU by therapies in the United States, in USD million (2019-2032)
- Table 34: Total Market Size of PKU in EU4 and the UK in USD million (2019-2032)
- Table 35: Market Size of PKU by therapies in EU4 and the UK, in USD million (2019-2032)
- Table 36: Total Market Size of PKU in Japan, in USD million (2019-2032)
- Table 37: Market Size of PKU by therapies in Japan, in USD million (2019-2032)
List of Figures
- Figure 1: Phenylketonuria
- Figure 2: Inheritance pattern
- Figure 3: Neurological damage in PKU
- Figure 4: Pathophysiology of PKU according to European guidelines
- Figure 5: Symptoms of PKU
- Figure 6: Newborn screening algorithm for PKU
- Figure 7: Diagnostic flow-chart for PKU
- Figure 8: Total Diagnosed Prevalent Cases of PKU in the 7MM (2019-2032)
- Figure 9: Total Diagnosed Prevalent Cases of PKU in the United States (2019-2032)
- Figure 10: Mutation type-specific Cases of PKU in the United States (2019-2032)
- Figure 11: Age-specific Cases of PKU in the United States (2019-2032)
- Figure 12: Severity-specific Cases of PKU in the United States (2019-2032)
- Figure 13: Total Diagnosed Prevalent Cases of PKU in EU4 and the UK (2019-2032)
- Figure 14: Mutation type-specific Cases of PKU in EU4 and the UK (2019-2032)
- Figure 15: Age-specific Cases of PKU in EU4 and the UK (2019-2032)
- Figure 16: Severity-specific Cases of PKU in EU4 and the UK (2019-2032)
- Figure 17: Total Diagnosed Prevalent Cases of PKU in Japan (2019-2032)
- Figure 18: Mutation type-specific Cases of PKU in Japan (2019-2032)
- Figure 19: Age-Specific Cases of PKU in Japan (2019-2032)
- Figure 20: Severity-specific Cases of PKU in Japan (2019-2032)
- Figure 21: Total Market Size of PKU in the 7MM, USD million (2019-2032)
- Figure 22: Market Size of PKU by Therapies in the 7MM, USD million (2019-2032)
- Figure 23: Total Market Size of PKU in the United States, USD million (2019-2032)
- Figure 24: Market size of PKU by therapies in the United States, in USD million (2019-2032)
- Figure 25: Total Market Size of PKU in EU4 and the UK, USD million (2019-2032)
- Figure 26: Market Size of PKU by therapies, in EU4 and the UK, in USD million (2019-2032)
- Figure 27: Total Market Size of PKU in Japan, USD million (2019-2032)
- Figure 28: Market Size of PKU by therapies in Japan, in USD million (2019-2032)
- Figure 29: Unmet needs
- Figure 30: Health Technology Assessment
- Figure 31: Reimbursement Process in Germany
- Figure 32: Reimbursement process in France
- Figure 33: Reimbursement process in Italy
- Figure 34: Reimbursement process in Spain
- Figure 35: Reimbursement process in the United Kingdom
- Figure 36: Reimbursement process in Japan
Key Highlights:
- Phenylketonuria (PKU) is an inborn metabolic defect that can be detected in early life days through routine newborn screening. PKU is defined by the absence or deficiency of an enzyme called phenylalanine hydroxylase (PAH), which is responsible for the amino acid phenylalanine processing.
- Newborn blood testing identifies almost all cases of phenylketonuria. All 50 states in the United States require newborns to be screened for PKU. Many other countries also routinely screen infants for PKU. If a person has PKU or family history, the doctor may recommend screening tests before pregnancy or birth. It is possible to identify PKU carriers through a blood test.
- According to DelveInsight estimates, in 2022, the highest number of diagnosed prevalent cases of Phenylketonuria (PKU) was observed in the United States among the 7MM.
- Most PKU cases are diagnosed in infants due to the high rate of newborn screening.
- The pipeline for PKU consists of promising drugs that are anticipated to change the treatment landscape of PKU. The current market anticipates the emergence of PTC923 (PTC Therapeutics) and SYNB1934 (Synlogic) products.
- In 2022, the market size of Phenylketonuria (PKU) was found to be ~610 million in the 7MM. The market size is estimated to increase during the forecast period (2023-2032).
DelveInsight's "Phenylketonuria (PKU) - Market Insights, Epidemiology, and Market Forecast - 2032" report delivers an in-depth understanding of the Phenylketonuria (PKU), historical and forecasted epidemiology as well as the Phenylketonuria (PKU) market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Phenylketonuria (PKU) market report provides current treatment practices, emerging drugs, market share of individual therapies, and current and forecasted 7MM Phenylketonuria (PKU) market size from 2019 to 2032. The report also covers current Phenylketonuria (PKU) treatment practices/algorithms and unmet medical needs to curate the best opportunities and assess the market's potential.
Geography Covered:
- The United States
- The EU4 (Germany, France, Italy, Spain) and the United Kingdom
- Japan
Study Period: 2019-2032
Phenylketonuria (PKU) Disease Understanding and Treatment Algorithm
Phenylketonuria (PKU) Overview
Phenylketonuria (PKU) is an inborn error of metabolism that is detectable during the first days of life via routine newborn screening. PKU is characterized by the absence or deficiency of an enzyme called phenylalanine hydroxylase (PAH), responsible for processing the amino acid phenylalanine. Amino acids are the chemical building blocks of proteins and are essential for proper growth and development. With normal PAH activity, phenylalanine is converted to another amino acid, tyrosine. However, when PAH is absent or deficient, phenylalanine accumulates and is toxic to the brain. Without treatment, most people with PKU would develop a severe intellectual disability. To prevent intellectual disability, treatment consists of a carefully controlled, phenylalanine-restricted diet beginning during the first days or weeks of life.
Phenylketonuria (PKU) Diagnosis
PKU diagnosis is achieved soon after birth by neonatal screening in most developed countries. In the countries where expanded newborn screening has been adopted, PKU is diagnosed by Phe and Tyr's evaluation in neonatal dried blood spot (DBS) using tandem mass spectrometry. Virtually 100% of PKU subjects should be identified. The risk of false-negative has become more pressing with early postnatal discharge from the hospital for mothers and term infants. In such cases, Phe/Tyr ratio evaluation increases the test's sensitivity and positive predictive value, avoiding the false-negative and lowering the false-positive risk. Although still utilized, other methods used for PKU neonatal screening do not allow the simultaneous evaluation of Tyr in neonatal DBS. Bacterial inhibition assay (Guthrie test) is a simple, inexpensive, and reliable test used for many decades; however, it is a manual, semi-quantitative test and is being replaced by other methods in all screening laboratories. Some laboratories use a fluorimetric test that is quantitative, automated, and reliable.
Note: Further details related to diagnosis are provided in the report.
Phenylketonuria (PKU) Treatment
PKU treatment aims to keep plasma phenylalanine levels within 120-360 mol/L (2-6 mg/dL), usually accomplished with a well-planned and regulated diet. Because phenylalanine is an essential amino acid, limiting the child's intake must be done cautiously. A healthy diet can help avoid intellectual disability and neurological, behavioral, and dermatological issues. Some intellectual dysfunction can be predicted if treatment is not initiated early; however, some late-treated youngsters have performed admirably. According to studies, children with PKU who are treated with a low phenylalanine diet before the age of 3 months fare well, with an IQ in the normal range. The special diet should start as soon as PKU is diagnosed and continue for the rest of the person's life. New medicines are being developed to lower PKU in the blood, but medical nutrition therapy is the main treatment for PKU. Treatment for PKU involves following a strict diet that is low in phenylalanine. A diet can prevent intellectual disability and neurological, behavioral, and dermatological problems. Babies with PKU need to be on a special formula as soon as possible, and it is advised to children and adults to eat a low-protein diet. Patients should avoid high-protein foods like milk, dairy, meats, eggs, nuts, soy, beans, and artificial sweeteners such as aspartame, which contains phenylalanine.
Phenylketonuria (PKU) Epidemiology
As the market is derived using the patient-based model, the Phenylketonuria (PKU) epidemiology chapter in the report provides historical as well as forecasted epidemiology segmented by total diagnosed prevalent cases of Phenylketonuria (PKU), mutation type-specific cases of Phenylketonuria (PKU), age-specific cases of Phenylketonuria (PKU), and severity-specific cases of Phenylketonuria (PKU) in the 7MM covering the United States, EU4 countries (Germany, France, Italy, and Spain) and the United Kingdom, and Japan from 2019 to 2032. The total diagnosed prevalent cases of Phenylketonuria (PKU) in the 7MM made up approximately 50,500 cases in 2022 and are projected to increase during the forecast period.
- The United States contributed to the largest diagnosed prevalent population of Phenylketonuria (PKU), acquiring ~36% of the 7MM in 2022.
- Among EU4 and the UK, Germany accounted for the highest number of Phenylketonuria (PKU) cases, followed by France, whereas Spain had the lowest number of cases in 2022.
- The mutation-specific cases of PKU were further divided into missense mutations, nonsense mutations, mutations at splice sites, deletions, and others. It was found that missense mutations formed the highest number of cases of PKU, whereas the others category formed the least number of cases of PKU, throughout the 7MM, in 2022.
- It was found that missense mutations formed the highest number of cases of PKU, i.e., ~11,300 cases, whereas the others category formed the least number of cases of PKU, i.e., ~850 cases in the US, in 2022.
Phenylketonuria (PKU) Drug Chapters
The drug chapter segment of the Phenylketonuria (PKU) report encloses a detailed analysis of Phenylketonuria (PKU)-marketed drugs and emerging (Phase-III and Phase II and Phase I/II) pipeline drugs. It also helps understand the Phenylketonuria (PKU) clinical trial details, expressive pharmacological actions, agreements and collaborations, approval and patent details, advantages and disadvantages of each included drug, and the latest news and press releases.
Current Therapies
KUVAN (Sapropterin Hydrochloride): Asubio-Pharma/BioMarin-Pharmaceutical
KUVAN is a synthetic form of BH4, the cofactor for the enzyme phenylalanine hydroxylase (PAH). PAH hydroxylates Phe through an oxidative reaction to form tyrosine. In patients with PKU, PAH activity is absent or deficient. Treatment with BH4 can activate residual PAH enzyme activity, improve the normal oxidative metabolism of Phe, and decrease Phe levels in some patients.
KUVAN is used in conjunction with a Phe-restricted diet to reduce blood phenylalanine (Phe) levels in patients with hyperphenylalaninemia (HPA) caused by tetrahydrobiopterin (BH4) responsive PKU. In January 2004, KUVAN was granted ODD by the FDA for the treatment of PKU. As far as the patent of KUVAN is concerned, BioMarin holds patents in Europe related to KUVAN that are valid until at least 2024. There are generic versions of KUVAN approved by the FDA, manufactured by Dr. Reddy's, Par Pharma, and Annora Pharma.
PALYNZIQ (pegvaliase-pqpz/rAvPAL-PEG/BMN 165): BioMarin Pharmaceutical
PALYNZIQ (pegvaliase-pqpz) injectable is the first FDA-approved enzyme replacement medicine for people with PKU who have uncontrolled blood Phe levels exceeding 600 mol/L (10 mg/dL) despite existing treatment. PALYNZIQ is a once-daily self-administered medication that works independently of the phenylalanine hydroxylase (PAH) enzyme, making it an option for all eligible adult PKU patients. In October 2020, the US FDA approved the supplemental biologics license application to increase the maximum allowable dose of 60 mg with PALYNZIQ injection for treating adults with PKU.
Note: Detailed current therapies assessment will be provided in the full Phenylketonuria (PKU) report.
Emerging Drugs
Sepiapterin (PTC923): PTC Therapeutics
PTC923 is an oral formulation of synthetic sepiapterin, a precursor to intracellular tetrahydrobiopterin, a key enzyme cofactor in the metabolism and synthesis of several metabolic products. Sepiapterin reductase is an enzyme involved in the manufacture of tetrahydrobiopterin, which has been shown in limited studies to regulate tumor growth.
Sepiapterin, a natural precursor of BH4, is a more stable molecule that is more readily transported across cellular membranes, giving it a potentially considerable advantage over BH4 as a treatment for disorders associated with BH4 deficiency. In May 2021, the United States Food and Drug Administration (FDA) and European Commission (EC) granted Orphan Drug Designation (ODD) for PTC923 to treat patients with hyperphenylalaninemia.
PTC923 has been shown to induce a larger increase in humans in circulating BH4 vs. sapropterin dihydrochloride. Sapropterin reduces blood phenylalanine (Phe) by =20-30% in a minority of subjects with PKU. This was according to a Phase II randomized, multicenter, three-period crossover, open-label, active-controlled, all-comers (regardless of phenylalanine hydroxylase [PAH] variants) comparison of PTC923 60 mg/kg, PTC923 20 mg/kg and sapropterin 20 mg/kg in 24 adults with phenylketonuria (PKU) and hyperphenylalaninemia.
This drug has a similar mechanism of action to the approved drug KUVAN and has better efficacy when compared to KUVAN due to greater mean change in Phe baseline levels.
SYNB1934: Synlogic
SYNB1934 is an orally administered, non-systemically absorbed drug candidate being studied as a potential biotherapeutic for phenylketonuria (PKU), an inherited metabolic disease marked by an inability to break down the amino acid phenylalanine (Phe), which can be neurotoxic. Lifelong, consistent control of low Phe levels is required to avoid PKU's serious risks and complications. Treatment options for PKU are currently limited, with a majority of individuals with PKU in need of treatment or not adequately responding to treatment.
SYNB1934 has a novel mechanism of action. The drug reduces levels of Phe in people with PKU by consuming Phe in the gastrointestinal (GI) tract, using genetic engineering of the well-characterized probiotic E. coli Nissle. Findings to date support the potential for an oral, efficacious, safe, convenient, and flexible treatment option for PKU.
Recently, in January 2023, SYNB1934 was granted Rare Pediatric Disease Designation (RPDD) by the US FDA for the potential treatment of phenylketonuria (PKU).
It was found that both the drugs (PTC923 and SYNB1934) had promising safety and efficacy according to the Phase II trials. The results of the Phase-III trials will further validate the safety and efficacy of these drugs.
Note: Detailed emerging therapies assessment will be provided in the final report.
Drug Class Insights
The existing Phenylketonuria treatment is mainly dominated by dietary supplements, a low-protein diet, and KUVAN, administered in conjugation with the supplements.
KUVAN belongs to the class of drugs called cofactors. It works by helping the body to break down phenylalanine so it will not build up in the blood. Clinical trials indicate that, depending on their PAH gene mutation, a subset of children with classic PKU respond to tetrahydrobiopterin (BH4) therapy. Sapropterin (synthetic BH4) is a cofactor for the enzyme phenylalanine hydroxylase. (PAH). Sapropterin is a synthetic version of BH4, which functions as a cofactor in the enzyme phenylalanine hydroxylase. (PAH). PAH hydroxylates phenylalanine (Phe) through an oxidative process to create tyrosine. Patients with PKU have low or no PAH activity. In some cases, BH4 treatment can activate remaining PAH, improve normal oxidative metabolism of Phe, and lower Phe levels. It should be combined with a Phe-restricted diet.
PALYNZIQ (pegvaliase) is an enzyme used to treat phenylketonuria in patients with phenylalanine levels that are too high on the current treatment. PEGylated phenylalanine ammonia-lyase (PAL) replaces poor phenylalanine hydroxylase (PAH) enzyme activity in PKU patients and lowers blood phenylalanine levels. Pegvaliase-pqpz is a homotetrameric protein of recombinant phenylalanine ammonia lyase (rAvPAL) and N-hydroxysuccinimide (NHS)-methoxypolyethylene glycol. (PEG).
Phenylketonuria (PKU) Market Outlook
Phenylketonuria (PKU) is an uncommon hereditary condition in which the body accumulates an amino acid called phenylalanine; amino acids are the building blocks of protein. Newborns with PKU do not have any symptoms at first; nevertheless, newborns commonly acquire signs of PKU without treatment within a few months. PKU patients can alleviate symptoms and avoid complications by following a particular diet and using drugs.
If treatment is not initiated early, some intellectual dysfunction can be predicted. The main strategy to treat PKU is following a diet restricting phenylalanine-containing foods; newborns with PKU may be fed breast milk. They must also take a particular formula called Lofenalac. A safe amount of phenylalanine varies from person to person with PKU and can change over time. The objective is not to ingest more than the phenylalanine required for optimal growth and bodily functioning. Because phenylalanine is found in almost all natural proteins, it is impossible to appropriately restrict the diet using only natural foods without jeopardizing health.
As a result, phenylalanine-free dietary preparations are beneficial. Protein-rich foods, such as meat, milk, fish, and cheese, are often prohibited from a diet. Low-protein foods, such as fruits, vegetables, and some grains, are naturally permitted in restricted quantities. People with PKU should also avoid the sweetener aspartame included in some meals, drinks, prescriptions, and supplements. When aspartame is digested, it releases phenylalanine, elevating the phenylalanine level in a person's blood.
Another supplement that could be added to the PKU diet is neutral amino acid treatment, which comes in powder or tablet form. This supplement may inhibit some phenylalanine absorption, which may be a therapy option for adults with PKU. Consult your doctor or a dietitian to see if this supplement is acceptable for your diet.
As mentioned earlier, KUVAN and PALYNZIQ are the approved drugs for treating Phenylketonuria (PKU). KUVAN is an oral pharmaceutical formulation of BH4, a natural cofactor for the PAH enzyme that enhances the residual PAH enzyme's activity in converting phenylalanine to tyrosine. KUVAN should be used in conjunction with a low-phenylalanine diet. PALYNZIQ is an injectable enzyme therapy for people on current medication who have uncontrolled blood phenylalanine levels.
Researchers and other scientists are investigating additional therapies for PKU. Two treatments are available for large neutral amino acid supplementation, which may help prevent phenylalanine from entering the brain, and enzyme replacement therapy, which employs a substance similar to the enzyme that normally breaks down phenylalanine.
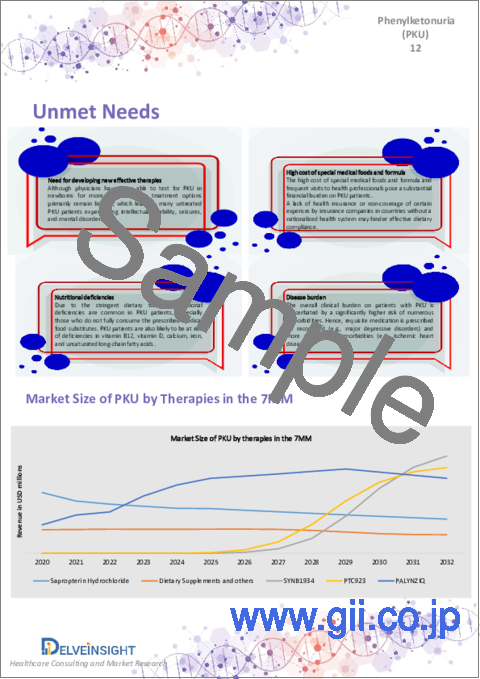
The treatment of Phenylketonuria (PKU) is multifaceted. The current market has been segmented based on the prevailing treatment pattern across the 7MM, which presents minor variations in the overall prescription pattern. Sapropterin Hydrochloride, dietary supplements, and PALYNZIQ have been considered in the forecast model.
The expected launch of upcoming therapies and greater integration of newborn screening in various countries, medication in secondary care and other clinical settings, research on best methods for implementation, and an upsurge in awareness will eventually facilitate the development of effective treatment options. However, there are a few barriers to treatment due to limited treatment options, resulting in many untreated Phenylketonuria patients experiencing intellectual disability, seizures, and mental disorders. With KUVAN limited to BH4-responsive patients and PALYNZIQ limited by its unfavorable safety and tolerability profile, more than 70% of PKU patients currently lack access to any form of medical treatment must rely solely on dietary restrictions and supplements. As a result, there is an urgent need for more focused and comprehensive disease management that incorporates the fundamental P4 medicine principles (predictive, preventive, personalized, and participatory).
A few players like PTC Therapeutics (PTC923), Synlogic (SYNB1934), and others are evaluating their lead candidates for the treatment of Phenylketonuria (PKU). These therapies are expected to change the Phenylketonuria (PKU) treatment landscape.
- The total market size of Phenylketonuria (PKU) in the United States was approximately USD 410 million in 2022 and is projected to grow during the forecast period (2023-2032).
- According to the estimates, Japan recorded the least market share, i.e., around 1% of the total market size of phenylketonuria in the 7MM in 2022.
- Among the EU4 (Germany, France, Italy, Spain) and the UK, Germany had the maximum revenue share in 2022, while Spain had the lowest market share.
- In 2022, sapropterin hydrochloride captured the highest market size of Phenylketonuria (PKU) in the 7MM.
Phenylketonuria (PKU) Drugs Uptake
This section focuses on the uptake rate of potential drugs expected to launch in the market during the forecast period (2023-2032). For example, for PTC923, we expect the drug uptake in the US to be medium with a probability-adjusted peak share of approximately 15%, and years to the peak is expected to be 7 years from the year of launch.
Note: Further detailed analysis of emerging therapies drug uptake in the report.
Phenylketonuria (PKU) Pipeline Development Activities
The report provides insights into different therapeutic candidates in Phase III, Phase II, and Phase I/II stage. It also analyzes key players involved in developing targeted therapeutics.
Pipeline Development Activities
The report covers detailed information on collaborations, acquisition and merger, licensing, and patent details for Phenylketonuria emerging therapies.
KOL Views
To keep up with current market trends, we take KOLs and SMEs' opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts contacted for insights on Phenylketonuria (PKU) evolving treatment landscape, patient reliance on conventional therapies, patient's therapy switching acceptability, drug uptake along with challenges related to accessibility, including Medical/scientific writers and Professors at the University of North Texas, Jichi Medical School, Medical professionals at Cruces University Hospital, Spain and Children's Hospital of Pittsburgh, US.
Delveinsight's analysts connected with 50+ KOLs to gather insights; however, interviews were conducted with 15+ KOLs in the 7MM. Centers such as Universitario Virgen del Rocio, Spain, Jichi Medical School, Japan, etc., were contacted. Their opinion helps understand and validate current and emerging therapies, treatment patterns, or Phenylketonuria (PKU) market trends. This will support the clients in potential upcoming novel treatments by identifying the overall scenario of the market and the unmet needs.
Qualitative Analysis
We perform Qualitative and market Intelligence analysis using various approaches, such as SWOT and Conjoint Analysis. In the SWOT analysis, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. These pointers are based on the Analyst's discretion and assessment of the patient burden, cost analysis, and existing and evolving treatment landscape.
Conjoint Analysis analyzes multiple approved and emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
For Phenylketonuria (PKU), the drug's efficacy was evaluated based on the percentage mean Phe (D5-Phe) reduction in plasma and fasting levels of plasma Phe. Further, the safety of the molecule is evaluated wherein the adverse events are majorly observed, and it sets a clear understanding of the side effects posed by the drug in the trials, directly affecting the safety of the molecule in the upcoming trials.
Market Access and Reimbursement
Reimbursement of rare disease therapies can be limited due to lack of supporting policies and funding, challenges of high prices, lack of specific approaches to evaluating rare disease drugs given limited evidence, and payers' concerns about budget impact. The high cost of rare disease drugs usually has a limited impact on the budget due to the small number of eligible patients being prescribed the drug. The US FDA has approved several rare disease therapies in recent years. From a patient perspective, health insurance and payer coverage guidelines surrounding rare disease treatments restrict broad access to these treatments, leaving only a small number of patients who can bypass insurance and pay for products independently.
Since 1987, NORD has given assistance programs to patients who could not afford life-saving or life-sustaining medication. Medication, financial aid with insurance premiums and copays, diagnostic testing assistance, and travel assistance for clinical trials or consultations with disease specialists are all available through these programs. The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc. NORD's PKU patient assistance programs provide financial assistance to eligible individuals diagnosed with PKU who have limited resources to pay for out-of-pocket healthcare costs.
Several companies have various reimbursement systems, strategies, and insurance policies to assist them in dealing with the prescription expense burden.
The KUVAN copay support program pays for all copay costs associated with KUVAN prescriptions, up to the annual maximum, for as long as the patient is in therapy and is eligible for treatment. The program may be terminated or modified at any moment. The PALYNZIQ copay assistance program also works similarly.
Scope of the Report:
- The report covers a segment of key events, an executive summary, and a descriptive overview of Phenylketonuria (PKU), explaining its causes, signs and symptoms, pathogenesis, and currently available therapies.
- Comprehensive insight into the epidemiology segments and forecasts, the future growth potential of diagnosis rate, disease progression, and treatment guidelines have been provided.
- Additionally, an all-inclusive account of the current and emerging therapies and the elaborative profiles of late-stage and prominent therapies will impact the current treatment landscape.
- A detailed review of the Phenylketonuria (PKU) market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies, by understanding trends, through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help shape and drive the 7MM Phenylketonuria (PKU) market.
Phenylketonuria (PKU) Report Insights
- Patient Population
- Therapeutic Approaches
- Phenylketonuria (PKU) Pipeline Analysis
- Phenylketonuria (PKU) Market Size and Trends
- Existing and Future Market Opportunity
Phenylketonuria (PKU) Report Key Strengths
- Ten Years Forecast
- The 7MM Coverage
- Phenylketonuria (PKU) Epidemiology Segmentation
- Key Cross Competition
- Attribute Analysis
- Drugs Uptake and Key Market Forecast Assumptions
Phenylketonuria (PKU) Report Assessment
- Current Treatment Practices
- Unmet Needs
- Pipeline Product Profiles
- Market Attractiveness
- Qualitative Analysis (SWOT and Conjoint Analysis)
Key Questions
Market Insights
- What was the Phenylketonuria (PKU) market share (%) distribution in 2019, and what would it look like in 2032? What are the contributing factors for this growth?
- How will the drug SYNB1934, with a novel mechanism of action, affect the treatment paradigm of Phenylketonuria (PKU)?
- What will be the impact of KUVAN generics on the Phenylketonuria (PKU) market in the near future?
- How will PTC923 compete with SYNB1934 in the Phenylketonuria (PKU) market?
- What are the pricing variations among different geographies for approved and off-label therapies?
- How would the market drivers, barriers, and future opportunities affect the market dynamics and subsequent analysis of the associated trends?
Epidemiology Insights
- What are the disease risk, burdens, and unmet needs of Phenylketonuria (PKU)? What will be the growth opportunities across the 7MM with respect to the patient population pertaining to Phenylketonuria (PKU)?
- What is the historical and forecasted Phenylketonuria (PKU) patient pool in the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan?
- Which type of Phenylketonuria (PKU), as per severity, is the largest contributor to the Phenylketonuria (PKU) patient pool?
- Which age group account for the major share of the Phenylketonuria (PKU) patient population? Or which age group is more susceptible to Phenylketonuria (PKU) in the 7MM?
Current Treatment Scenario and Emerging Therapies
- What are the current options for the treatment of Phenylketonuria (PKU)? What are the current guidelines for treating Phenylketonuria (PKU) in the US and Europe?
- How many companies are developing therapies for treating Phenylketonuria (PKU)?
- How many emerging therapies are in the mid-stage and late stage of development for treating Phenylketonuria (PKU)?
- What are the recent novel therapies, targets, mechanisms of action, and technologies developed to overcome the limitation of existing therapies?
- What are the key designations that have been granted for emerging therapies for Phenylketonuria (PKU)?
- What is the cost burden of approved therapies on the patient?
- What is the patient acceptability in terms of preferred treatment options as per real-world scenarios?
- What are the country-specific accessibility issues of expensive, recently approved therapies?
- What is the 7MM historical and forecasted market of Phenylketonuria (PKU)?
Reasons to buy:
- The report will help develop business strategies by understanding the latest trends and changing treatment dynamics driving the Phenylketonuria (PKU) market.
- Insights on patient burden/disease prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- To understand the existing market opportunity in varying geographies and the growth potential over the coming years.
- Distribution of historical and current patient share based on real-world prescription data along with reported sales of approved products in the US, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan.
- Identifying strong upcoming players in the market will help devise strategies that will help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the Conjoint analysis section to provide visibility around leading classes.
- Highlights of access and reimbursement policies of approved therapies, barriers to accessibility of expensive off-label therapies, and patient assistance programs.
- To understand Key Opinion Leaders' perspectives around the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet need of the existing market so that the upcoming players can strengthen their development and launch strategy
Table of Contents
1. Key Insights
2. Report Introduction
3. Phenylketonuria (PKU) Market Overview at a Glance
- 3.1. Market Share (%) Distribution of Phenylketonuria (PKU) in 2019
- 3.2. Market Share (%) Distribution of Phenylketonuria (PKU) in 2032
4. Executive Summary of Phenylketonuria (PKU)
5. Key Events
6. Disease Background and Overview
- 6.1. Introduction
- 6.2. Inheritance of PKU
- 6.3. Classification of PKU
- 6.4. Pathophysiology of PKU
- 6.5. Symptoms of PKU
- 6.6. Prognosis of PKU
- 6.7. Diagnosis of PKU
- 6.7.1. Bacterial Inhibition Test
- 6.7.2. Tandem Mass Spectrometry
- 6.7.3. Molecular Analysis of PKU-associated PAH Mutations
- 6.7.4. Genotype-phenotype Correlation
- 6.8. Differential Diagnosis
- 6.9. Diagnosis Guidelines
- 6.9.1. ACMG Practice Guidelines
- 6.9.2. Key European Guidelines for the Diagnosis and Management of Patients With PKU
- 6.10. Treatment
- 6.10.1. Approved Drugs of PKU
- 6.10.2. Key European Guidelines for the Diagnosis and Management of Patients With Phenylketonuria (2017)
- 6.10.3. Phenylalanine Hydroxylase Deficiency: Diagnosis and Management Guideline (ACMG Practice Guidelines 2014)
- 6.10.4. Medical and Dietary Guidelines for the Treatment of PKU (developed by the American College of Medical Genetics and Genomics (ACMG) and Genetic Metabolic Dietician's International [GMDI])
7. Methodology
8. Epidemiology and Patient Population
- 8.1. Key Findings
- 8.2. Assumptions and Rationale
- 8.3. Epidemiology Scenario: The 7MM
- 8.3.1. 7MM Total Diagnosed Prevalent Cases of PKU
- 8.4. The United States
- 8.4.1. Total Diagnosed Prevalent Cases of PKU in the United States
- 8.4.2. Mutation type-specific Cases of PKU in the United States
- 8.4.3. Age-specific Cases of PKU in the United States
- 8.4.4. Severity-specific Cases of PKU in the United States
- 8.5. EU4 and the UK
- 8.5.1. Total Diagnosed Prevalent Cases of PKU in EU4 and the UK
- 8.5.2. Mutation type-specific Cases of PKU in EU4 and the UK
- 8.5.3. Age-specific Cases of PKU in EU4 and the UK
- 8.5.4. Severity-specific Cases of PKU in EU4 and the UK
- 8.6. Japan
- 8.6.1. Total Diagnosed Prevalent Cases of PKU in Japan
- 8.6.2. Mutation type-specific Cases of PKU in Japan
- 8.6.3. Age-specific Cases of PKU in Japan
- 8.6.4. Severity-specific Cases of PKU in Japan
9. Patient Journey
10. Marketed Drugs
- 10.1. Key-cross Competition
- 10.2. KUVAN (Sapropterin Hydrochloride): Asubio-Pharma/BioMarin-Pharmaceutical
- 10.2.1. Product Description
- 10.2.2. Regulatory Milestones
- 10.2.3. Other Developmental Activities
- 10.2.4. Clinical Developmental Activities
- 10.2.5. Safety and Efficacy
- 10.3. PALYNZIQ (pegvaliase-pqpz/rAvPAL-PEG/BMN 165): BioMarin Pharmaceutical
- 10.3.1. Product Description
- 10.3.2. Regulatory Milestones
- 10.3.3. Other Developmental Activities
- 10.3.4. Clinical Developmental Activities
- 10.3.5. Safety and Efficacy
11. Emerging Drugs
- 11.1. Key Cross Competition
- 11.2. Sepiapterin (PTC923): PTC Therapeutics
- 11.2.1. Product Description
- 11.2.2. Other Developmental Activities
- 11.2.3. Clinical Developmental Activities
- 11.2.4. Safety and Efficacy
- 11.3. SYNB1934: Synlogic
- 11.3.1. Drug Description
- 11.3.2. Other Developmental Activities
- 11.3.3. Clinical Developmental Activities
- 11.3.4. Safety and Efficacy
12. Phenylketonuria (PKU): The 7MM Analysis
- 12.1. Key Findings
- 12.2. Key Market Forecast Assumptions
- 12.3. Attribute Analysis
- 12.4. The 7MM Market Outlook
- 12.5. The 7MM Size: PKU
- 12.5.1. Total Market size of PKU in the 7MM
- 12.5.2. Market Size of PKU by Therapies in the 7MM
- 12.6. Market Size of PKU in the United States
- 12.6.1. Total Market Size of PKU in the United States
- 12.6.2. Market Size of PKU by Therapies in the United States
- 12.7. Market Size of PKU in EU4 and the UK
- 12.7.1. Total Market Size of PKU in EU4 and the UK
- 12.7.2. Market Size of PKU by Therapies in EU4 and the UK
- 12.8. Market Size of Phenylketonuria in Japan
- 12.8.1. Total Market Size of PKU in Japan
- 12.8.2. Market Size of PKU by Therapies in Japan
13. KOL Views
14. SWOT Analysis
15. Unmet Needs
16. Market Access and Reimbursement
- 16.1. The United States
- 16.1.1. Centre for Medicare and Medicaid Services (CMS)
- 16.2. EU4 and the UK
- 16.2.1. Germany
- 16.2.2. France
- 16.2.3. Italy
- 16.2.4. Spain
- 16.2.5. United Kingdom
- 16.3. Japan
- 16.3.1. MHLW
17. Appendix
- 17.1. Bibliography
- 17.2. Report Methodology