|
|
市場調査レポート
商品コード
1179470
エプスタインバーウイルス(EBV) - 市場考察、疫学、市場予測(2032年)Epstein Barr Virus (EBV) - Market Insight, Epidemiology and Market Forecast - 2032 |
||||||
|
● お客様のご希望に応じて、既存データの加工や未掲載情報(例:国別セグメント)の追加などの対応が可能です。 詳細はお問い合わせください。 |
|||||||
| エプスタインバーウイルス(EBV) - 市場考察、疫学、市場予測(2032年) |
|
出版日: 2022年12月01日
発行: DelveInsight
ページ情報: 英文 170 Pages
納期: 1~3営業日
|
- 全表示
- 概要
- 図表
- 目次
当レポートでは、エプスタインバーウイルス(EBV)の主要7市場(米国・ドイツ・フランス・イタリア・スペイン・英国・日本)について調査分析し、市場規模と予測、現在の治療法と新薬の情報などを提供しています。
目次
第1章 主要考察
第2章 レポートのイントロダクション
第3章 エプスタインバーウイルス(EBV)市場の概要
- エプスタインバーウイルス(EBV)の市場シェア分布(2019年)
- エプスタインバーウイルス(EBV)の市場シェア分布(2032年)
第4章 エプスタインバーウイルス(EBV)のエグゼクティブサマリー
第5章 主な出来事
第6章 疫学と市場調査手法
第7章 疾患の背景と概要
- イントロダクション
- EBVの遺伝子型と株変異
- 臨床症状
- 病因と危険因子
- 病態生理学
- EBVに関連する合併症
- 診断
- 管理
- 臨床ガイドライン
第8章 疫学と患者数
- 主な調査結果
- 前提条件の根拠
- 主要7市場の診断されたEBV-IMの総発生例
- 主要7市場のEBV陽性がんとPTLDの総発生例
- 米国
- 欧州4ヶ国・英国
- 日本
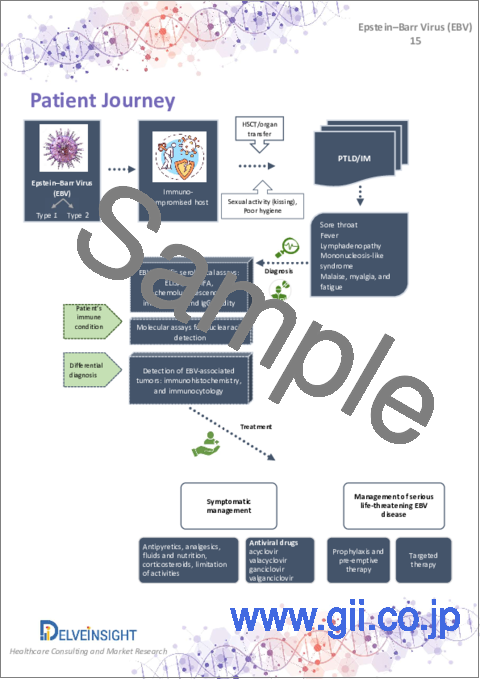
第9章 ペイシェントジャーニー
第10章 新たな治療法
第11章 エプスタインバーウイルス(EBV):主要7市場の分析
- 主な調査結果
- 主要7市場のEBVの総市場規模
- 市場の見通し
- EBVワクチン
- EBV陽性がん/PTLD
- 主な市場の予測の前提条件
- 米国の市場規模
- 米国のEBVの市場規模
- 米国のEBV-IMの市場規模:治療法別
- 米国のEBV陽性がんとPTLDの市場規模(1L)
- 米国のEBV陽性がんとPTLD(R/R)の市場規模
- 欧州4ヶ国・英国の市場規模
- 欧州4ヶ国・英国のEBVの総市場規模
- 欧州4ヶ国・英国のEBV-IMの市場規模:治療法別
- 欧州4ヶ国・英国のEBV陽性がんとPTLDの市場規模(1L)
- 欧州4ヶ国・英国のEBV陽性がんとPTLD(R/R)の市場規模
- 日本の市場規模
- 日本のEBVの市場規模
- 日本のEBV-IMの市場規模:治療法別
- 日本のEBV陽性がんとPTLDの市場規模(1L)
- 日本のEBV陽性がんとPTLD(R/R)の市場規模
第12章 SWOT分析
第13章 KOLの見解
第14章 アンメットニーズ
第15章 市場参入
第16章 付録
第17章 DelveInsightのサービス内容
第18章 免責事項
第19章 DelveInsightについて
List of Tables
- Table 1: Summary of EBV, Market, Epidemiology, and Key Events (2019-2032)
- Table 2: Key Events
- Table 3: Diagnostic Methods for EBV Detection
- Table 4: Advantages and Disadvantages of Various EBV Diagnostic Methods
- Table 5: Differential Diagnosis
- Table 6: ECIL-6 Scoring System
- Table 7: Major Sources Used to Derive Cancer and Transplantation Cases
- Table 8: Total Diagnosed Incident Cases of EBV-IM in the 7MM (2019-2032)
- Table 9: Total Incident Cases of EBV+ Cancers and PTLD in the 7MM (2019-2032)
- Table 10: Total Diagnosed Incident Cases of EBV-IM in the US (2019-2032)
- Table 11: Incident Cases of EBV+ Cancers in the US (2019-2032)
- Table 12: Incident Cases of EBV+ PTLD in the US (2019-2032)
- Table 13: Diagnosed Incident cases of EBV-IM in EU4 and the UK (2019-2032)
- Table 14: Incident Cases of EBV+ Cancers in EU4 and the UK (2019-2032)
- Table 15: Incident Cases of EBV+ PTDL in EU4 and the UK (2019-2032)
- Table 16: Diagnosed Incident Cases of EBV-IM in Japan (2019-2032)
- Table 17: Incident Cases of EBV+ Cancers in Japan (2019-2032)
- Table 18: Incident Cases of EBV+ PTLD in Japan (2019-2032)
- Table 19: Key cross of the key emerging drugs
- Table 20: Nanatinostat in Combination With valganciclovir, Clinical Trial Description, 2022
- Table 21: EBVALLO (tabelecleucel), Clinical Trial Description, 2022
- Table 22: Posoleucel (ALVR-105) Clinical Trial Description, 2022
- Table 23: Total Market Size of EBV in the 7MM in USD million (2019-2032)
- Table 24: Key Market Forecast Assumptions for Nana-val
- Table 25: Key Market Forecast Assumptions for tabelecleucel
- Table 26: Key Market Forecast Assumptions for posoleucel
- Table 27: Market Size of EBV in the United States in USD million (2019-2032)
- Table 28: Market Size of EBV-IM in the United States by Therapies in USD million (2019-2032)
- Table 29: Market Size of EBV+ Cancer and PTLD (1L) in the United States in USD million (2019-2032)
- Table 30: Market Size of EBV+ Cancer and PTLD (R/R) in the United States in USD million (2019-2032)
- Table 31: Total Market Size of EBV in EU4 and the UK in USD million (2019-2032)
- Table 32: Market Size of EBV-IM in EU4 and the UK by Therapies in USD million (2019-2032)
- Table 33: Market Size of EBV+ Cancer and PTLD (1L) in EU4 and USD million (2019-2032)
- Table 34: Market Size of EBV+ Cancer and PTLD (R/R) in EU4 and the UK in USD million (2019-2032)
- Table 35: Market Size of EBV in Japan in USD million (2019-2032)
- Table 36: Market Size of EBV-IM in Japan by Therapies in USD million (2019-2032)
- Table 37: Market Size of EBV+ Cancer and PTLD (1L) in Japan in USD million (2019-2032)
- Table 38: Market Size of EBV+ Cancer and PTLD (R/R) in Japan in USD million (2019-2032)
List of Figures
- Figure 1: Clinical Manifestations of EBV infection
- Figure 2: Main Symptoms of IM
- Figure 3: Risk Factors Associated with EBV Infection
- Figure 4: EBV Infection in Healthy Carriers
- Figure 5: Pathogenesis of HSCT-PTLD
- Figure 6: Symptomatic Management of IM
- Figure 7: Flowchart of the EBV Protocol
- Figure 8: Total Diagnosed Incident Cases of EBV-IM in the 7MM (2019-2032)
- Figure 9: Total Incident Cases of EBV+ Cancers and PTLD in the 7MM (2019-2032)
- Figure 10: Total Diagnosed Incident Cases of EBV-IM in the US (2019-2032)
- Figure 11: Incident Cases of EBV+ Cancers in the US (2019-2032)
- Figure 12: Incident Cases of EBV+ PTLD in the US (2019-2032)
- Figure 13: Diagnosed Incident Cases of EBV-IM in EU4 and the UK (2019-2032)
- Figure 14: Incident Cases of EBV+ Cancers in EU4 and the UK (2019-2032)
- Figure 15: Incident Cases of EBV+ PTDL in EU4 and the UK (2019-2032)
- Figure 16: Diagnosed Incident Cases of EBV-IM in Japan (2019-2032)
- Figure 17: Incident Cases of EBV+ Cancers in Japan (2019-2032)
- Figure 18: Incident Cases of EBV+ PTLD in Japan (2019-2032)
- Figure 19: Approach of Nana-val Targeting and Killing EBV+ Cancer Cells
- Figure 20: Total Market Size of EBV in the 7MM (2019-2032)
- Figure 21: Market Size of EBV in the United States (2019-2032)
- Figure 22: Market Size of EBV-IM in the United States by Therapies (2019-2032)
- Figure 23: Market Size of EBV+ Cancer and PTLD (1L) in the United States by Therapies (2019-2032)
- Figure 24: Market Size of EBV+ Cancer and PTLD (R/R) in the United States (2019-2032)
- Figure 25: Total Market Size of EBV in EU4 and the UK (2019-2032)
- Figure 26: Market Size of EBV-IM in EU4 and the UK by Therapies (2019-2032)
- Figure 27: Market Size of EBV+ Cancer and PTLD (1L) in EU4 and the UK (2019-2032)
- Figure 28: Market Size of EBV+ Cancer and PTLD (R/R) in EU4 and the UK (2019-2032)
- Figure 29: Market Size of EBV in Japan (2019-2032)
- Figure 30: Market Size of EBV-IM in Japan by Therapies (2019-2032)
- Figure 31: Market Size of EBV+ Cancer and PTLD (1L) in Japan (2019-2032)
- Figure 32: Market Size of EBV+ Cancer and PTLD (R/R) in Japan (2019-2032)
DelveInsight's 'Epstein-Barr Virus - Market Insights, Epidemiology, and Market Forecast - 2032' report delivers an in-depth understanding of the historical and forecasted epidemiology as well as the market trends of Epstein-Barr Virus in the United States, EU4 (Germany, Spain, Italy, France) and the United Kingdom, and Japan.
The Epstein-Barr Virus market report provides current treatment practices, emerging drugs, market share of individual therapies, and current and forecasted Epstein-Barr Virus market size from 2019 to 2032, segmented by the 7MM. The report also covers the current Epstein-Barr Virus treatment practice/algorithm, market drivers, market barriers, and unmet medical needs to curate the best opportunities and assesses the underlying market potential.
Geographies Covered
- The United States
- EU4 and the United Kingdom
- Japan
Study period: 2019-2032
Epstein-Barr Virus: Disease Understanding and Treatment Algorithm
Epstein-Barr Virus Overview
Epstein-Barr Virus (EBV) is a common virus that remains dormant in most people. EBV causes infectious mononucleosis (IM) and has been associated with certain cancers, including Burkitt lymphoma, immunoblastic lymphoma, nasopharyngeal cancer, and stomach (gastric) cancer.
The transmission of EBV occurs in several ways, such as deep kissing or food-sharing. Increased levels of viral DNA are found in salivary secretions after the initial infection. Children can be infected after eating food that an EBV-infected individual has already chewed. The transmission has occurred through stem cells, organ transplantation, and blood transfusion.
The clinical presentation of the EBV infection usually overlaps with other acute viral syndromes caused by other viruses, such as Cytomegalovirus (CMV) and hepatitis viruses, which can lead to similar symptoms. Diagnostic schemes of EBV differ according to the patient's immune condition because the importance and urgency of therapeutic intervention differ between immunocompromised and immunocompetent individuals; a wide range of assays was utilized in diagnosing EBS.
Therapeutic strategies include prophylaxis, preemptive therapy, and targeted therapy. Rituximab, reduction of immunosuppression, and EBV-specific cytotoxic T-cell therapy are recommended as first-line therapy, whilst unselected donor lymphocyte infusions or chemotherapy are options as second-line therapy; other methods, including antiviral drugs, are discouraged.
Epstein-Barr Virus Diagnosis and Treatment
It covers the details of conventional and current medical therapies and diagnoses available in the Epstein-Barr Virus market to treat the condition. It also provides country-wise treatment guidelines and algorithms across the United States, Europe, and Japan.
Epstein-Barr Virus Epidemiology
The Epstein-Barr Virus epidemiology division provides insights into the historical and current patient pool and the forecasted trend for the 7MM. It helps recognize the causes of current and forecasted trends by exploring numerous studies and views of KOL. The report also provides the prevalent patient pool, trends, and assumptions.
Key Findings
The disease epidemiology covered in the report provides historical and forecasted Epstein-Barr Virus epidemiology segmented as the Diagnosed Incident Cases of Epstein-Barr Virus IM, Incident Cases of Epstein-Barr Virus + Cancer, Incident cases of Epstein-Barr Virus+ PTLD. The report includes the total Epstein-Barr Virus cases in the 7MM covering the United States, EU4 and the United Kingdom, and Japan from 2019 to 2032.
Country-wise Epstein-Barr Virus Epidemiology
The epidemiology segment also provides the Epstein-Barr Virus epidemiology data and findings across the United States, EU4 and the United Kingdom, and Japan.
The total number of Epstein-Barr Virus IM cases in the 7MM countries was more than 1,120,000 in 2021.
As per the estimates, the United States had the highest number of incident cases of Epstein-Barr Virus in 2021. Among EU4 and the UK, Germany had the highest number of Epstein-Barr Virus cases, with more than 146,000 cases, followed by France in 2021. On the other hand, Spain had the lowest number of Epstein-Barr Virus cases, with close to 79,000 cases in 2021.
Epstein-Barr Virus Drug Chapters
The drug chapter segment of the Epstein-Barr Virus report encloses a detailed analysis of Epstein-Barr Virus-marketed drugs and late-stage (Phase III, Phase II/III, and Phase II) pipeline drugs. It also helps understand the Epstein-Barr Virus clinical trial details, expressive pharmacological action, agreements and collaborations, approval and patent details, advantages and disadvantages of each included drug, and the latest news and press releases.
Treatment generally supports mononucleosis; antipyretics and anti-inflammatory medications help treat fever, sore throat, and general fatigue in this illness. Hydration, rest, and good nutritional intake should also be encouraged. Corticosteroids are not generally recommended in the routine treatment of mononucleosis because of concerns with immunosuppression; however, in cases of airway obstruction, corticosteroids are indicated along with appropriate airway management.
Products detail in the report…
Epstein-Barr Virus Emerging Drugs
Drug developers are gradually shifting their attention toward Epstein-Barr Virus to meet the patient pool's current demands and counter the unmet needs of the therapeutic market.
Many molecules are in the pipeline to treat Epstein-Barr Virus-associated infection patients across many countries to cater to their needs. Some of the major late-stage products are likely to hit the market during our study period 2019-2032 include tabelecleucel (Atara Biotherapeutics), posoleucel (AlloVir), and Nanatinostat + valganciclovir (Viracta Therapeutics).
Tabelecleucel: Atara Biotherapeutics
Tabelecleucel (Atara Biotherapeutics) is an off-the-shelf, allogeneic T-cell immunotherapy based on Atara Biotherapeutics' allogeneic T-cell immunotherapy platform. The drug has received BTD in the US, EMA Priority Medicines (PRIME) in Europe, and ODD in the US and EU for treatment of EBV+ PTLD following allogeneic hematopoietic cell transplant (HCT). The drug is currently in the late clinical phases for potentially treating patients with rituximab-refractory EBV+ PTLD. The therapy has received a marketing authorization recommendation from EMA in the EU for EBVALLO (tabelecleucel) to treat adult and pediatric patients who experience a serious complication following solid organ transplantation (SOT) or HCT called EBV+ PTLD.
Posoleucel (ALVR105): Allovir
Posoleucel (ALVR105), being investigated by AlloVir, is a multivirus specific T-cell therapy (VST) targeting five devastating viral pathogens: BK virus (BKV), Cytomegalovirus (CMV), Adenovirus (AdV), EBV, and human herpesvirus 6 (HHV-6), that can transform care for transplant recipients as well as individuals who are at high risk for opportunistic viral infections by reducing or preventing disease morbidity and dramatically improving patient outcomes. The drug has received the ODD, PRIME designation in the EU, and RMAT designation from the US for treating serious infections with BKV, CMV, HHV-6, EBV, and/or AdV in allogeneic HSCT recipients. The therapy is currently being investigated in Phase II and III trials and is expected to get launched in the US market by 2024.
Products detail in the report…
Epstein-Barr Virus Market Outlook
The Epstein-Barr Virus market outlook of the report builds a detailed comprehension of the historical, current, and forecasted Epstein-Barr Virus market trends by analyzing the impact of current therapies on the market, unmet needs, and demand for better technology.
This segment gives a thorough detail of the Epstein-Barr Virus market trend of each marketed drug and late-stage pipeline therapy by evaluating their impact based on the annual cost of therapy, inclusion and exclusion criteria, mechanism of action, compliance rate, growing need for the market, increasing patient pool, covered patient segment, expected launch year, competition with other therapies, brand value, their impact on the market, and KOL view. The calculated market data are presented with relevant tables and graphs to give a clear view of the market at first sight.
As per DelveInsight, the Epstein-Barr Virus market in the 7MM is expected to change in 2019-2032.
Key Findings
This section includes a glimpse of the Epstein-Barr Virus market in the 7MM. The Epstein-Barr Virus market size in the seven major markets was approximately USD 3,100 million in 2021.
The United States: Market Outlook
This section provides the total Epstein-Barr Virus market size and the market size of Epstein-Barr Virus by therapies in the United States.
The United States accounts for the largest Epstein-Barr Virus market size than EU4 and the United Kingdom, and Japan.
EU4 and the UK: Market Outlook
This section provides the total Epstein-Barr Virus market size. It also provides Epstein-Barr Virus market size by therapies in Germany, France, Italy, Spain, and the United Kingdom.
Japan Market Outlook
This section provides the total Epstein-Barr Virus market size. It also provides the market size of Epstein-Barr Virus by therapies in Japan.
Epstein-Barr Virus Drugs Uptake
This section focuses on the uptake rate of potential drugs launched or expected to be launched in the market during 2019-2032. The analysis covers the Epstein-Barr Virus market uptake by drugs, patient uptake by therapies, and sales of each drug.
This helps in understanding the drugs with the most rapid uptake and the reasons behind the maximal use of new drugs and allows the comparison of the drugs based on market share and size, which again will be useful in investigating factors important in the market uptake and making financial and regulatory decisions.
Epstein-Barr Virus Pipeline Development Activities
The report provides insights into different therapeutic candidates in Phase III, Phase II/III, Phase II, and Phase I/II. It also analyses Epstein-Barr Virus's key players in developing targeted therapeutics.
Major players include Atara Biotherapeutics and Allovir, whose key products are expected to get launched in the US market by 2028.
Pipeline Development Activities
The report covers collaborations, acquisitions, mergers, licensing, and patent details for emerging Epstein-Barr Virus therapies.
KOL Views
To keep up with current market trends, we take KOLs and SMEs' opinions working in the Epstein-Barr Virus domain through primary research to fill the data gaps and validate our secondary research. Their opinion helps understand and validate current and emerging treatment patterns or Epstein-Barr Virus market trends. This will support the clients in potential novel treatments by identifying the overall scenario of the market and the unmet needs.
Competitive Intelligence Analysis
We perform a competitive and market intelligence analysis of the Epstein-Barr Virus market using various competitive intelligence tools: SWOT analysis, PESTLE analysis, Porter's five forces, BCG matrix, market entry strategies, etc. The inclusion of the analysis entirely depends upon the data availability.
Scope of the Report:
- The report covers a descriptive overview of the Epstein-Barr Virus, explaining its causes, signs and symptoms, pathophysiology, and currently available therapies.
- Comprehensive insight is provided into the Epstein-Barr Virus epidemiology and treatment in the 7MM.
- An all-inclusive account of the current and emerging therapies for Epstein-Barr Virus is provided, along with the assessment of new therapies that will impact the current treatment landscape.
- A detailed review of the Epstein-Barr Virus market, historical and forecasted, is included in the report, covering drug outreach in the 7MM.
- The report provides an edge while developing business strategies by understanding trends shaping and driving the global Epstein-Barr Virus market.
Report Highlights:
- Recently, the Epstein-Barr Virus market has been set to change due to the increase in investment and research funding for Epstein-Barr Virus treatment, and the survival rates of those with Epstein-Barr Virus have increased because of advancements in treatment, such as blood and bone marrow stem cell transplants; which would expand the market size to enable the drug manufacturers to penetrate more into the market.
- The companies and academics are working to assess challenges and seek opportunities that could influence Epstein-Barr Virus R&D. The therapies under development are focused on novel approaches to treat/improve the disease condition.
- Major players are involved in developing therapies for Epstein-Barr Virus. Launching emerging therapies will significantly impact the Epstein-Barr Virus market.
- For Epstein-Barr Virus, a better understanding of disease pathogenesis will also contribute to developing novel therapeutics.
- Our in-depth analysis of the pipeline assets across different stages of development (Phase III and Phase II), different emerging trends, and comparative analysis of pipeline products with detailed clinical profiles, key cross-competitor, launch date along with product development activities will support the clients in the decision-making process regarding their therapeutic portfolio by identifying the overall scenario of the R&D activities.
Epstein-Barr Virus Report Insights
- Patient Population
- Therapeutic Approaches
- Epstein-Barr Virus Pipeline Analysis
- Epstein-Barr Virus Market Size and Trends
- Market Opportunities
- Impact of Upcoming Therapies
Epstein-Barr Virus Report Key Strengths
- 11-year Forecast
- The 7MM Coverage
- Epstein-Barr Virus Epidemiology Segmentation
- Key Competitors
- Highly Analyzed Market
- Drugs Uptake
Epstein-Barr Virus Report Assessment
- Current Treatment Practices
- Unmet Needs
- Pipeline Product Profiles
- Market Attractiveness
Key Questions
Market Insights
- What was the market share percentage distribution, and how would it look by 2032?
- What would be the total market size and market size of Epstein-Barr Virus by therapies across the 7MM forecast period (2019-2032)?
- What are the key findings about the market across the 7MM, and which country will have the largest Epstein-Barr Virus market size during the forecast period (2019-2032)?
- At what CAGR is the Epstein-Barr Virus market expected to grow in the 7MM forecast period (2019-2032)?
- What would be the Epstein-Barr Virus market outlook across the 7MM forecast period (2019-2032)?
- What would be the Epstein-Barr Virus market growth until 2032 and the resultant market size by 2032?
- How would future opportunities affect the market dynamics and subsequent analysis of the associated trends?
Epidemiology Insights
- What are the disease risk, burdens, and unmet needs of Epstein-Barr Virus?
- What is the historical patient pool of Epstein-Barr Virus covering the US, EU4 and the UK, and Japan?
- What would be the forecasted patient pool of Epstein-Barr Virus covering the US, EU4 and the UK, and Japan?
- What will be the growth opportunities in the 7MM concerning the patient population of Epstein-Barr Virus?
- Out of all the 7MM, which country would have the highest prevalence of Epstein-Barr Virus during the forecast period (2019-2032)?
- At what CAGR is the population expected to grow in the 7MM forecast period (2019-2032)?
Current Treatment Scenario, Marketed Drugs, and Emerging Therapies
- What are the current options to treat Epstein-Barr Virus?
- What are the current treatment guidelines for treating Epstein-Barr Virus in the US, Europe, and Japan?
- What are the Epstein-Barr Virus-marketed drugs and their MOA, regulatory milestones, product development activities, advantages, disadvantages, safety, efficacy, etc.?
- How many companies are developing therapies to treat Epstein-Barr Virus?
- How many therapies are developed by each company to treat Epstein-Barr Virus?
- How many emerging therapies are in the mid-stage and late stages of development to treat Epstein-Barr Virus?
- What are the key collaborations (Industry-Industry, Industry-Academia), Mergers and acquisitions, and licensing activities related to Epstein-Barr Virus therapies?
- What are the recent novel therapies, targets, mechanisms of action, and technologies developed to overcome the limitation of existing therapies?
- What are the clinical studies for Epstein-Barr Virus and its status?
- Which key designations have been granted for the emerging therapies for Epstein-Barr Virus?
- What are the global historical and forecasted Epstein-Barr Virus markets?
Reasons to Buy:
- The report will help develop business strategies by understanding trends shaping and driving the Epstein-Barr Virus market.
- To understand the future market competition in the Epstein-Barr Virus market and an insightful review of the key market drivers and barriers.
- Organize sales and marketing efforts by identifying the best opportunities for Epstein-Barr Virus in the US, EU4 (Germany, Spain, Italy, France) and the UK, and Japan.
- Identifying upcoming solid players in the market will help devise strategies that will help get ahead of competitors.
- Organize sales and marketing efforts by identifying the best opportunities for the Epstein-Barr Virus market.
- To understand the future market competition in the Epstein-Barr Virus market.
Table of Contents
1. Key Insights
2. Report Introduction
3. Epstein-Barr Virus (EBV) Market Overview at a Glance
- 3.1. Market Share (%) Distribution of Epstein-Barr Virus (EBV) in 2019
- 3.2. Market Share (%) Distribution of Epstein-Barr Virus (EBV) in 2032
4. Executive Summary of Epstein-Barr Virus (EBV)
5. Key Events
6. Epidemiology and Market Methodology
7. Disease Background and Overview
- 7.1. Introduction
- 7.2. EBV Genotypes and Strain Variation
- 7.3. Clinical Manifestations
- 7.4. Etiology and Risk Factors
- 7.5. Pathophysiology
- 7.5.1. Pathogenesis of PTLD after HSCT due to EBV
- 7.6. Complications Associated with EBV
- 7.7. Diagnosis
- 7.7.1. Differential diagnosis
- 7.8. Management
- 7.8.1. Symptomatic management of IM
- 7.8.2. Management of serious or life-threatening EBV disease
- 7.8.3. Treatment of EBV-related lymphomas
- 7.8.4. Prevention
- 7.9. Clinical Guidelines
- 7.9.1. Management of EBV Infections and PTLD in Patients after Allogeneic HSCT: Sixth European Conference on Infections in Leukemia (ECIL-6) Guidelines
- 7.9.2. Post-transplant Lymph Proliferative Disorders, Epstein-Barr Virus Infection, and Disease in Solid Organ Transplantation: Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice
- 7.9.3. Front-line Management of Post-transplantation Lymphoproliferative Disorder in Adult Solid Organ Recipient Patients - A British Society for Hematology Guideline
8. Epidemiology and Patient Population
- 8.1. Key Findings
- 8.2. Assumption Rationale
- 8.3. Total Diagnosed Incident Cases of EBV-IM in the 7 MM
- 8.4. Total Incident Cases of EBV+ Cancers and PTLD in the 7 MM
- 8.5. The US
- 8.5.1. Diagnosed Incident Cases of EBV-IM in the US
- 8.5.2. Incident Cases of EBV+ Cancers in the US
- 8.5.3. Incident Cases of EBV+ PTLD in the US
- 8.6. EU4 and the UK
- 8.6.1. Diagnosed Incident Cases of EBV-IM in EU4 and the UK
- 8.6.2. Incident Cases of EBV+ Cancers in EU4 and the UK
- 8.6.3. Incident Cases of EBV+ PTLD in EU4 and the UK
- 8.7. Japan
- 8.7.1. Diagnosed Incident Cases of EBV-IM in Japan
- 8.7.2. Incident Cases of EBV+ Cancers in Japan
- 8.7.3. Incident Cases of EBV+ PTLD in Japan
9. Patient Journey
10. Emerging Therapies
- 10.1. Key Cross Competition
- 10.2. Nanatinostat (VRx-3996) in Combination With valganciclovir: Viracta Therapeutics
- 10.2.1. Drug description
- 10.2.2. Other development activities
- 10.2.3. Clinical development
- 10.2.4. Clinical trial information
- 10.2.5. Safety and efficacy
- 10.2.6. Product profile
- 10.2.7. Analyst comment
- 10.3. EBVALLO (tabelecleucel): Atara Biotherapeutics
- 10.3.1. Drug description
- 10.3.2. Other development activities
- 10.3.3. Clinical development
- 10.3.3.1. Clinical trial information
- 10.3.4. Safety and efficacy
- 10.3.5. Product profile
- 10.3.6. Analyst comment
- 10.4. Posoleucel (viralym-M; ALVR-105): AlloVir
- 10.4.1. Drug description
- 10.4.2. Other development activities
- 10.4.3. Clinical development
- 10.4.4. Clinical trial information
- 10.4.5. Safety and efficacy
- 10.4.6. Product profile
- 10.4.7. Analyst comment
11. Epstein-Barr Virus (EBV): The 7MM Analysis
- 11.1. Key Findings
- 11.2. Total Market Size of EBV in the 7MM
- 11.3. Market Outlook
- 11.3.1. EBV-vaccine
- 11.3.2. EBV+ Cancers/PTLD
- 11.4. Key Market Forecast Assumptions
- 11.5. The US Market Size
- 11.5.1. Market Size of EBV in the United States
- 11.5.2. Market Size of EBV-IM by Therapies in the United States
- 11.5.3. Market Size of EBV+ Cancer and PTLD in the United States (1L)
- 11.5.4. Market Size of EBV+ Cancer and PTLD (R/R) in the United States
- 11.6. EU4 and the UK Market Size
- 11.6.1. Total Market Size of EBV in EU4 and the UK
- 11.6.2. Market Size of EBV-IM by Therapies in EU4 and the UK
- 11.6.3. Market Size of EBV+ Cancer and PTLD in EU4 and the UK (1L)
- 11.6.4. Market Size of EBV+ Cancer and PTLD (R/R) in EU4 and the UK
- 11.7. Japan Market Size
- 11.7.1. Market Size of EBV in Japan
- 11.7.2. Market Size of EBV-IM by Therapies in Japan
- 11.7.3. Market Size of EBV+ Cancer and PTLD in Japan (1L)
- 11.7.4. Market Size of EBV+ Cancer and PTLD (R/R) in Japan
12. SWOT Analysis
13. KOL Views
14. Unmet Need
15. Market Access
- 15.1. Global Disease Burden
- 15.2. US
- 15.2.1. Medicare
- 15.2.2. Long-term disability for Epstein-Barr Virus
- 15.2.3. EBV mononucleosis testing coverage
- 15.3. Europe
- 15.4. Japan
16. Appendix
- 16.1. Bibliography
- 16.2. Acronyms and Abbreviations
- 16.3. Report Methodology