|
|
市場調査レポート
商品コード
1275636
体外診断の世界市場:提供別、技術別、用途別、診断アプローチ別 - 予測(~2030年)In-Vitro Diagnostics Market by Offering (Kits, Software), Technology (Immunoassay, Molecular Diagnostics, Rapid Tests, Biochemistry), Application (Infectious Diseases, Oncology), Diagnostic Approach (Lab, POC)-Global Forecast to 2030 |
||||||
|
● お客様のご希望に応じて、既存データの加工や未掲載情報(例:国別セグメント)の追加などの対応が可能です。 詳細はお問い合わせください。 |
|||||||
| 体外診断の世界市場:提供別、技術別、用途別、診断アプローチ別 - 予測(~2030年) |
|
出版日: 2023年05月17日
発行: Meticulous Research
ページ情報: 英文 526 Pages
納期: 即納可能
|
- 全表示
- 概要
- 図表
- 目次
世界の体外診断の市場規模は、2030年までに1,301億米ドルに達し、2023年~2030年にCAGRで4.6%の成長が予測されています。市場の成長は、主に高齢者人口の増加に伴う慢性疾患の有病率の上昇、急性と慢性の感染症の流行、研究活動資金の増加、疾患の早期診断に関する意識の高まり、ポイントオブケア(PoC)診断や迅速診断の需要の高まり、医療支出の増加、研究活動資金の増加などに起因しています。
当レポートでは、世界の体外診断市場について調査分析し、市場動向の考察、地域とセグメントの評価、競合情勢などを提供しています。
目次
第1章 概要
第2章 調査手法
第3章 エグゼクティブサマリー
第4章 市場の考察
- 概要
- 促進要因
- 抑制要因
- 機会
- 課題
- 技術動向
第5章 体外診断市場:規制分析
- 概要
- 北米
- 米国
- カナダ
- 欧州
- アジア太平洋
- 中国
- 日本
- インド
- ラテンアメリカ
- 中東
第6章 体外診断市場の評価:提供別
- 概要
- 試薬・キット
- システム
- ソフトウェア・サービス
第7章 体外診断市場の評価:技術別
- 概要
- 分子診断
- ポリメラーゼ連鎖反応(PCR)
- ハイブリダイゼーション
- 等温核酸増幅技術
- DNAシーケンシング・次世代シーケンシング
- マイクロアレイ
- 質量分析
- その他の分子診断技術
- ポイントオブケア(PoC)診断
- ラテラルフローアッセイ/迅速検査
- PoC分子診断
- その他のPoC製品
- イムノアッセイ/免疫化学
- 酵素結合免疫吸着測定法(ELISA)
- 酵素免疫スポットアッセイ(ELISPOT)
- ウェスタンブロッティング
- ラジオイムノアッセイ
- 生化学/臨床化学
- 代謝パネル
- 電解質パネル
- 肝臓パネル
- 脂質プロファイル
- 腎臓プロファイル
- 甲状腺機能パネル
- 全血グルコースモニタリング
- 微生物学
- 血液学
- 凝固/止血
- 尿検査
- その他のIVD技術
第8章 体外診断市場:用途別
- 概要
- 感染症
- COVID-19検査
- 性感染症(STD)検査
- 医療関連感染(HAI)
- 肝炎
- HIV
- 熱帯病
- インフルエンザ
- 呼吸器感染症(インフルエンザを除く)
- その他の感染症
- 腫瘍
- 糖尿病
- 心臓
- 腎臓
- 自己免疫疾患
- その他の用途
第9章 体外診断市場の評価:診断アプローチ別
- 概要
- 臨床検査
- OTC/セルフテスト
- ポイントオブケア検査
第10章 体外診断市場:エンドユーザー別
- 概要
- 病院・診療所
- 診断研究所
- 在宅医療
- その他のエンドユーザー
第11章 体外診断市場の評価:地域別
- 概要
- 北米
- 米国
- カナダ
- 欧州
- ドイツ
- フランス
- 英国
- イタリア
- スペイン
- スイス
- その他の欧州
- アジア太平洋
- 日本
- 中国
- インド
- 韓国
- オーストラリア
- その他のアジア太平洋
- ラテンアメリカ
- ブラジル
- メキシコ
- アルゼンチン
- その他のラテンアメリカ
- 中東
- サウジアラビア
- その他の中東
- アフリカ
- 南アフリカ
- その他のアフリカ
第12章 競合情勢
- 概要
- 主な成長戦略
- 競合ダッシュボード
- 業界のリーダー
- 市場の差別化要因
- 先導企業
- 新興企業
- ベンダーの市場ポジショニング
- 市場シェア分析(2022年:上位15社)
- F. Hoffman-La Roche Ltd. (Switzerland)
- Abbott Laboratories (U.S.)
- Danaher Corporation (U.S)
- Siemens Healthineers AG (Germany)
- Thermo Fisher Scientific Inc. (U.S.)
第13章 企業プロファイル
- Abbott Laboratories
- Becton, Dickinson and Company
- biomerieux S.A.
- Danaher Corporation
- F. Hoffman-La Roche Ltd
- Qiagen N.V.
- Siemens Healthineers AG (A Subsidiary of Siemens AG)
- Thermo Fisher Scientific Inc.
- Bio-Rad Laboratories, Inc.
- Wama Diagnostica
- lllumina, Inc.
- Wiener Laboratories SAIC
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd
- QuidelOrtho Corporation
- Agilent Technologies, Inc.
- DiaSorin S.p.A.
第14章 付録
List of Tables
- Table 1 Global IVD Market: Impact Analysis of Market Drivers (2023-2030)
- Table 2 Number of People With Diabetes Aged 20-79, by Region, 2021 Vs. 2030 Vs. 2045 (In Thousands)
- Table 3 Population Aged 65 Years Or Above, by Region, 2019 Vs. 2030 Vs. 2050 (In Millions)
- Table 4 Estimated Number of New Cancer Cases, by Type (2020 Vs. 2030)
- Table 5 HIV Statistics (2021)
- Table 6 Healthcare Expenditure As A Percentage of GDP, by Country (2014 Vs. 2019)
- Table 7 Healthcare Expenditure, by Country (2020, 2021, and 2022)
- Table 8 Global IVD Market: Impact Analysis of Market Restraints (2023-2030)
- Table 9 Requirements for CLIA-Waived and Moderate/High-complexity PoC Molecular Diagnostic Tests
- Table 10 Global IVD Market: Impact Analysis of Market Opportunities (2023-2030)
- Table 11 Emerging Countries: Percentage of Urban Population (2021 Vs. 2025 Vs. 2050)
- Table 12 Biomarkers and Corresponding Targeted Therapies Currently Available
- Table 13 Clinical Applications of Genomics and Proteomics in Infectious Disease Diagnosis
- Table 14 Global IVD Market: Impact Analysis of Market Challenges (2023-2030)
- Table 15 Regulatory Authorities Governing In Vitro Diagnostics, by Country
- Table 16 Some of the Emergency Use Authorizations (EUAs) Granted for IVD During the COVID-19 Pandemic
- Table 17 Global In Vitro Diagnostics (IVD) Market, by Offering, 2021-2030 (USD Million)
- Table 18 Global IVD Kits & Reagents Market, by Country/Region, 2021-2030 (USD Million)
- Table 19 Global IVD Systems Market, by Country/Region, 2021-2030 (USD Million)
- Table 20 Global IVD Software & Services Market, by Country/Region, 2021-2030 (USD Million)
- Table 21 Global In Vitro Diagnostics Market, by Technology, 2021-2030 (USD Million)
- Table 22 Global Molecular Diagnostics Market, by Type, 2021-2030 (USD Million)
- Table 23 Global Molecular Diagnostics Market, by Country/Region, 2021-2030 (USD Million)
- Table 24 Key Companies Offering PCR Products
- Table 25 Global Polymerase Chain Reaction Market, by Country/Region, 2021-2030 (USD Million)
- Table 26 Global Hybridization Market, by Country/Region, 2021-2030 (USD Million)
- Table 27 Key Companies Offering Isothermal Nucleic Acid Amplification Products
- Table 28 Global Isothermal Nucleic Acid Amplification Market, by Country/Region, 2021-2030 (USD Million)
- Table 29 Key Companies Offering DNA Sequencing & Next-generation Sequencing Products
- Table 30 Global DNA Sequencing and Next-Generation Sequencing Market, by Country/Region, 2021-2030 (USD Million)
- Table 31 Key Companies Offering Microarray Products
- Table 32 Global Microarrays Market, by Country/Region, 2021-2030 (USD Million)
- Table 33 Global Mass Spectrometry Market, by Country/Region, 2021-2030 (USD Million)
- Table 34 Other Molecular Diagnostic Technologies Market, by Country/Region, 2021-2030 (USD Million)
- Table 35 Global Point-of-Care (PoC) Diagnostics Market, by Technology, 2021-2030, (USD Million)
- Table 36 Global Point-of-Care (PoC) Diagnostics Market, by Country/Region, 2021-2030, (USD Million)
- Table 37 Global Lateral Flow Assays/Rapid Tests Market, by 2021-2030 (USD Million)
- Table 38 Global PoC Molecular Diagnostics Market, 2021-2030 (USD Million)
- Table 39 Other PoC Products Market, by Country/Region, 2021-2030 (USD Million)
- Table 40 Global Immunoassay/Immunochemistry Market, by Type, 2021-2030 (USD Million)
- Table 41 Global Immunoassay/Immunochemistry Market, by Country/Region, 2021-2030 (USD Million)
- Table 42 Global Enzyme-Linked Immunosorbent Assays (ELISA) Market, by Country/Region, 2021-2030 (USD Million)
- Table 43 ELISPOT Vs. Conventional ELISA
- Table 44 Global Enzyme-Linked Immunospot Assays (ELISPOT) Market, by Country/Region, 2021-2030 (USD Million)
- Table 45 Global Western Blotting Market, by Country/Region, 2021-2030 (USD Million)
- Table 46 Global Radioimmunoassay Market, by Country/Region, 2021-2030 (USD Million)
- Table 47 Global Biochemistry/Clinical Chemistry Market, by Type, 2021-2030 (USD Million)
- Table 48 Global Metabolic Panels Market, by Country/Region, 2021-2030 (USD Million)
- Table 49 Global Electrolyte Panels Market, by Country/Region, 2021-2030, (USD Million)
- Table 50 Global Liver Panels Market, by Country/Region, 2021-2030, (USD Million)
- Table 51 Global Lipid Profiles Market, by Country/Region, 2021-2030 (USD Million)
- Table 52 Global Renal Profiles Market, by Country/Region, 2021-2030 (USD Million)
- Table 53 Global Thyroid Function Panels Market, by Country/Region, 2021-2030 (USD Million)
- Table 54 Number of People With Diabetes Aged 20-79 Years, by Region, 2019 Vs. 2030 Vs. 2045 (In Million)
- Table 55 Global Whole Blood Glucose Monitoring Market, by Country/Region, 2021-2030 (USD Million)
- Table 56 Key Companies Offering Microbiology-based IVD Products
- Table 57 Global Microbiology Market, by Country/Region, 2021-2030 (USD Million)
- Table 58 Key Companies Offering Hematology Products
- Table 59 Global Hematology Market, by Country/Region, 2021-2030 (USD Million)
- Table 60 Key Companies Offering Coagulation and Hemostasis Products
- Table 61 Global Coagulation and Hemostasis Market, by Country/Region, 2021-2030 (USD Million)
- Table 62 Key Companies Offering Urinalysis Products
- Table 63 Global Urinalysis Market, by Country/Region, 2021-2030 (USD Million)
- Table 64 Global Other IVD Technologies Market, by Country/Region, 2021-2030 (USD Million)
- Table 65 Global In Vitro Diagnostics Market, by Application, 2021-2030 (USD Million)
- Table 66 Global In Vitro Diagnostics Market for Infectious Diseases, by Type, 2021-2030 (USD Million)
- Table 67 Global In Vitro Diagnostics Market for Infectious Diseases, by Country/Region, 2021-2030 (USD Million)
- Table 68 Recently Launched and Approved Rapid Diagnostic Tests for COVID-19 Detection
- Table 69 Global Invitro Diagnostics Market for COVID-19 Testing, by Country/Region, 2021-2030 (USD Million)
- Table 70 Global In Vitro Diagnostics Market for Sexually Transmitted Diseases (STD) Testing, by Country/Region, 2021-2030 (USD Million)
- Table 71 Global In Vitro Diagnostics Market for Healthcare-associated Infections (HAIs), by Country/Region, 2021-2030 (USD Million)
- Table 72 Global In Vitro Diagnostics Market for Hepatitis, by Country/Region, 2021-2030 (USD Million)
- Table 73 Global Prevalence of HIV, by Region (2020)
- Table 74 Global In Vitro Diagnostics Market for HIV, by Country/Region, 2021-2030 (USD Million)
- Table 75 Global In Vitro Diagnostics Market for Tropical Diseases, by Country/Region, 2021-2030 (USD Million)
- Table 76 Global In Vitro Diagnostics Market for Influenza, by Country/Region, 2021-2030 (USD Million)
- Table 77 Global In Vitro Diagnostics Market for Respiratory Infections, by Country/Region, 2021-2030 (USD Million)
- Table 78 Global In Vitro Diagnostics Market for Other Infectious Diseases, by Country/Region, 2021-2030 (USD Million)
- Table 79 Global In Vitro Diagnostics Market for Oncology, by Country/Region, 2021-2030 (USD Million)
- Table 80 Number of People With Diabetes Aged 20-79 Years, by Region, 2021 Vs. 2030 Vs. 2045 (In Million)
- Table 81 Global In Vitro Diagnostics Market for Diabetes, by Country/Region, 2021-2030 (USD Million)
- Table 82 Global In Vitro Diagnostics Market for Cardiology, by Country/Region, 2021-2030 (USD Million)
- Table 83 Global In Vitro Diagnostics Market for Nephrology, by Country/Region, 2021-2030 (USD Million)
- Table 84 Global In Vitro Diagnostics Market for Autoimmune Disorders, by Country/Region, 2021-2030 (USD Million)
- Table 85 Global In Vitro Diagnostics Market for Other Applications, by Country/Region, 2021-2030 (USD Million)
- Table 86 Global In Vitro Diagnostics Market, by Diagnostic Approach 2021-2030 (USD Million)
- Table 87 Global In Vitro Diagnostics Market for Laboratory Testing, by Country/Region, 2021-2030 (USD Million)
- Table 88 Global In Vitro Diagnostics Market for OTC/Self-testing, by Country/Region, 2021-2030 (USD Million)
- Table 89 Global In Vitro Diagnostics Market for Point-of-Care Testing, by Country/Region, 2021-2030 (USD Million)
- Table 90 Global In Vitro Diagnostics Market, by End User, 2021-2030 (USD Million)
- Table 91 Global In Vitro Diagnostics Market for Hospitals & Clinics, by Country/Region, 2021-2030 (USD Million)
- Table 92 Global In Vitro Diagnostics Market for Diagnostic Laboratories, by Country/Region, 2021-2030 (USD Million)
- Table 93 Global In Vitro Diagnostics Market for Home Healthcare, by Country/Region, 2021-2030 (USD Million)
- Table 94 Global In Vitro Diagnostics Market for Other End Users, by Country/Region, 2021-2030 (USD Million)
- Table 95 Global In Vitro Diagnostics Market, by Country/Region, 2021 Vs. 2030 (USD Million)
- Table 96 North America: In Vitro Diagnostics Market, by Country, 2021-2030 (USD Million)
- Table 97 North America: In Vitro Diagnostics Market, by Offering, 2021-2030 (USD Million)
- Table 98 North America: In Vitro Diagnostics Market, by Technology, 2021-2030 (USD Million)
- Table 99 North America: In Vitro Diagnostics Market for Immunoassay/Immunochemistry, by Type, 2021-2030 (USD Million)
- Table 100 North America: In Vitro Diagnostics Market for Biochemistry/Clinical Chemistry, by Type, 2021-2030 (USD Million)
- Table 101 North America: In Vitro Diagnostics Market for Molecular Diagnostics, by Type, 2021-2030 (USD Million)
- Table 102 North America: In Vitro Diagnostics Market for Point-of-Care (PoC) Diagnostics, by Type, 2021-2030 (USD Million)
- Table 103 North America: In Vitro Diagnostics Market, by Application, 2021-2030 (USD Million)
- Table 104 North America: In Vitro Diagnostics Market for Infectious Diseases, by Type, 2021-2030 (USD Million)
- Table 105 North America: In Vitro Diagnostics Market, by Diagnostics Approach, 2021-2030 (USD Million)
- Table 106 North America: In Vitro Diagnostics Market, by End User, 2021-2030 (USD Million)
- Table 107 U.S.: In Vitro Diagnostics Market, by Offering, 2021-2030 (USD Million)
- Table 108 U.S.: In Vitro Diagnostics Market, by Technology, 2021-2030 (USD Million)
- Table 109 U.S.: In Vitro Diagnostics Market for Immunoassay/Immunochemistry, by Type, 2021-2030 (USD Million)
- Table 110 U.S.: In Vitro Diagnostics Market for Biochemistry/Clinical Chemistry, by Type, 2021-2030 (USD Million)
- Table 111 U.S.: In Vitro Diagnostics Market for Molecular Diagnostics, by Type, 2021-2030 (USD Million)
- Table 112 U.S.: In Vitro Diagnostics Market for Point-of-Care (PoC) Diagnostics, by Type, 2021-2030 (USD Million)
- Table 113 U.S.: In Vitro Diagnostics Market, by Application, 2021-2030 (USD Million)
- Table 114 U.S.: In Vitro Diagnostics Market for Infectious Diseases, by Type, 2021-2030 (USD Million)
- Table 115 U.S.: In Vitro Diagnostics Market, by Diagnostics Approach, 2021-2030 (USD Million)
- Table 116 U.S.: In Vitro Diagnostics Market, by End User, 2021-2030 (USD Million)
- Table 117 Canada: Prevalence of Diseases/Conditions in the Geriatric (Age 65 Years and Above) Population (2019-2020)
- Table 118 Canada: In Vitro Diagnostics Market, by Offering, 2021-2030 (USD Million)
- Table 119 Canada: In Vitro Diagnostics Market, by Technology, 2021-2030 (USD Million)
- Table 120 Canada: In Vitro Diagnostics Market for Immunoassay/Immunochemistry, by Type, 2021-2030 (USD Million)
- Table 121 Canada: In Vitro Diagnostics Market for Biochemistry/Clinical Chemistry, by Type, 2021-2030 (USD Million)
- Table 122 Canada: In Vitro Diagnostics Market for Molecular Diagnostics, by Type, 2021-2030 (USD Million)
- Table 123 Canada: In Vitro Diagnostics Market for Point-of-Care (PoC) Diagnostics, by Type, 2021-2030 (USD Million)
- Table 124 Canada: In Vitro Diagnostics Market, by Application, 2021-2030 (USD Million)
- Table 125 Canada: In Vitro Diagnostics Market for Infectious Diseases, by Type, 2021-2030 (USD Million)
- Table 126 Canada: In Vitro Diagnostics Market, by Diagnostics Approach, 2021-2030 (USD Million)
- Table 127 Canada: In Vitro Diagnostics Market, by End User, 2021-2030 (USD Million)
- Table 128 Europe: In Vitro Diagnostics Market, by Country/Region, 2021-2030 (USD Million)
- Table 129 Europe: In Vitro Diagnostics Market, by Offering, 2021-2030 (USD Million)
- Table 130 Europe: In Vitro Diagnostics Market, by Technology, 2021-2030 (USD Million)
- Table 131 Europe: In Vitro Diagnostics Market for Immunoassay/Immunochemistry, by Type, 2021-2030 (USD Million)
- Table 132 Europe: In Vitro Diagnostics Market for Biochemistry/Clinical Chemistry, by Type, 2021-2030 (USD Million)
- Table 133 Europe: In Vitro Diagnostics Market for Molecular Diagnostics, by Type, 2021-2030 (USD Million)
- Table 134 Europe: In Vitro Diagnostics Market for Point-of-Care (PoC) Diagnostics, by Type, 2021-2030 (USD Million)
- Table 135 Europe: In Vitro Diagnostics Market, by Application, 2021-2030 (USD Million)
- Table 136 Europe: In Vitro Diagnostics Market for Infectious Diseases, by Type, 2021-2030 (USD Million)
- Table 137 Europe: In Vitro Diagnostics Market, by Diagnostics Approach, 2021-2030 (USD Million)
- Table 138 Europe: In Vitro Diagnostics Market, by End User, 2021-2030 (USD Million)
- Table 139 Germany: In Vitro Diagnostics Market, by Offering, 2021-2030 (USD Million)
- Table 140 Germany: In Vitro Diagnostics Market, by Technology, 2021-2030 (USD Million)
- Table 141 Germany: In Vitro Diagnostics Market for Immunoassay/Immunochemistry, by Type, 2021-2030 (USD Million)
- Table 142 Germany: In Vitro Diagnostics Market for Biochemistry/Clinical Chemistry, by Type, 2021-2030 (USD Million)
- Table 143 Germany: In Vitro Diagnostics Market for Molecular Diagnostics, by Type, 2021-2030 (USD Million)
- Table 144 Germany: In Vitro Diagnostics Market for Point-of-Care (PoC) Diagnostics, by Type, 2021-2030 (USD Million)
- Table 145 Germany: In Vitro Diagnostics Market, by Application, 2021-2030 (USD Million)
- Table 146 Germany: In Vitro Diagnostics Market for Infectious Diseases, by Type, 2021-2030 (USD Million)
- Table 147 Germany: In Vitro Diagnostics Market, by Diagnostics Approach, 2021-2030 (USD Million)
- Table 148 Germany: In Vitro Diagnostics Market, by End User, 2021-2030 (USD Million)
- Table 149 France: In Vitro Diagnostics Market, by Offering, 2021-2030 (USD Million)
- Table 150 France: In Vitro Diagnostics Market, by Technology, 2021-2030 (USD Million)
- Table 151 France: In Vitro Diagnostics Market for Immunoassay/Immunochemistry, by Type, 2021-2030 (USD Million)
- Table 152 France: In Vitro Diagnostics Market for Biochemistry/Clinical Chemistry, by Type, 2021-2030 (USD Million)
- Table 153 France
List of Figures
- Figure 1 Research Process
- Figure 2 Secondary Sources Referenced for This Study
- Figure 3 Primary Research Techniques
- Figure 4 Key Executives Interviewed
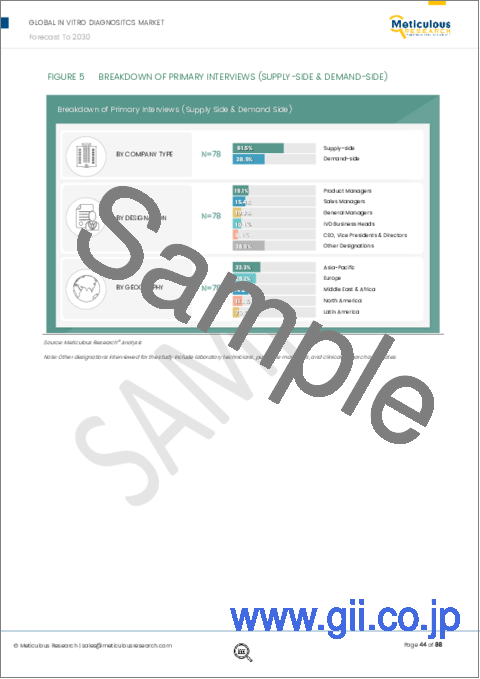
- Figure 5 Breakdown of Primary Interviews (Supply Side & Demand Side)
- Figure 6 Market Size Estimation
- Figure 7 Global In Vitro Diagnostics (IVD) Market, by Offering, 2023 Vs. 2030 (USD Million)
- Figure 8 Global in Vitro Diagnostic (IVD) Market, by Technology, 2023 Vs. 2030 (USD Million)
- Figure 9 Global in Vitro Diagnostic Market, by Application, 2023 Vs. 2030 (USD Million)
- Figure 10 Global in Vitro Diagnostic Market, by Diagnostic Approach, 2023 Vs. 2030 (USD Million)
- Figure 11 Global in Vitro Diagnostic Market, by End User, 2023 Vs. 2030 (USD Million)
- Figure 12 Global IVD Market, by Geography
- Figure 13 Factors Affecting Market Growth
- Figure 14 U.S.: Medical and Health R&D Expenditure, 2016-2020 (USD Billion)
- Figure 15 Global Biopharmaceutical R&D Expenditure, 2016-2028 (USD Billion)
- Figure 16 USFDA Regulatory Pathways for IVD Kits
- Figure 17 EU Regulatory Pathway - IVDR 2017/746
- Figure 18 China: Medical Device Classification and Pre-Market Requirements for IVD Devices
- Figure 19 Global In Vitro Diagnostics (IVD) Market, by Offering, 2023 Vs. 2030 (USD Million)
- Figure 20 Global in Vitro Diagnostic Market, by Technology, 2023 Vs. 2030 (USD Million)
- Figure 21 Global In Vitro Diagnostics Market, by Application, 2023 Vs. 2030 (USD Million)
- Figure 22 U.S.: Estimated Acute Hepatitis C Cases, 2011-2020
- Figure 23 Estimated Number of New Cancer Cases, by Region, 2020-2040 (Million)
- Figure 24 Total Number of Accredited Testing Laboratories (ISO/IEC 17025), 2016-2020
- Figure 25 Prevalence of Diabetes Across the World (In Million)
- Figure 26 Global In Vitro Diagnostics Market, by Diagnostic Approach 2023 Vs. 2030 (USD Million)
- Figure 27 Benefits of Laboratory Testing Approach
- Figure 28 Benefits of the Point-of-Care Testing Approach
- Figure 29 Global In Vitro Diagnostics Market, by End User, 2023 Vs. 2030 (USD Million)
- Figure 30 Global In Vitro Diagnostics Market, by Region, 2023 Vs. 2030 (USD Million)
- Figure 31 North America: Number of People Aged 65 Years and Above, 2015-2021 (Million)
- Figure 32 North America: Per Capita Health Expenditure (USD), 2017-2021
- Figure 33 North America: In Vitro Diagnostics Market Snapshot
- Figure 34 Canada: Number of People Aged 65 and Above, 2018-2022 (Million)
- Figure 35 Europe: Number of People Aged 65 and Above (In Million) (2015-2021)
- Figure 36 Europe: In Vitro Diagnostics Market Snapshot
- Figure 37 France: Percentage of Population Aged 65 Years and Above (2016-2021)
- Figure 38 France: Prevalence of Chronic Diseases in People Aged 65 and Above (2020)
- Figure 39 U.K: Number of Cancer Cases, 2010-2030 (Million)
- Figure 40 U.K.: Number of Diabetes and Urogenital, Blood, and Endocrine Disease Cases - DALYS (Disability-Adjusted Life Years), 2017-2020 (Thousand)
- Figure 41 U.K.: Number of People Diagnosed With Sexually Transmitted Infections, 2018-2020 (Thousand)
- Figure 42 Prevalence of HAI in Healthcare-Associated Infections in Acute-Care Wards (2016-2021)
- Figure 43 Italy: Ovarian Cancer Cases Diagnosed (2018-2020)
- Figure 44 Number of HIV Tests Performed in Catalonia (Spain) (2014-2021)
- Figure 45 Switzerland: Number of New Cancer Cases (2020-2040)
- Figure 46 Switzerland: Number of People Aged 65 Years and Above (2017-2021)
- Figure 47 Asia-Pacific: In Vitro Diagnostics Market Snapshot
- Figure 48 Japan: Percentage Share of the Geriatric Population (Aged 65 Years And Above) (2015-2022)
- Figure 49 Japan: New Cancer Cases Reported (2020-2040)
- Figure 50 China: Independent Medical Laboratories Market, 2015-2021 (USD Million)
- Figure 51 India: Number of People Aged 65 Years and Above, 2017-2021 (Million)
- Figure 52 Latin America: In Vitro Diagnostics Market Snapshot
- Figure 53 Brazil: Challenges To the In Vitro Diagnostics Market
- Figure 54 Middle East: In Vitro Diagnostics Market Snapshot
- Figure 55 Saudi Arabia: Number of People Aged 65 Years and Above, 2017-2021 (Thousand)
- Figure 56 Africa: In Vitro Diagnostics Market Snapshot
- Figure 57 South Africa: Number of People (All Ages) Living With HIV, 2010-2020 (Million)
- Figure 58 South Africa: Number of People With Diabetes, 2000-2045 (Thousand)
- Figure 59 Key Growth Strategies Adopted by Leading Players (2019-2023)
- Figure 60 Competitive Dashboard: Global In Vitro Diagnostics Market
- Figure 61 Global In Vitro Diagnostics Market Competitive Benchmarking, by Offering
- Figure 62 Market Share Analysis: Global In Vitro Diagnostics Market (2022)
- Figure 63 Abbott Laboratories: Financial Overview (2021)
- Figure 64 Becton, Dickinson and Company: Financial Overview (2022)
- Figure 65 biomerieux S.A.: Financial Overview (2021)
- Figure 66 Danaher Corporation: Financial Overview (2021)
- Figure 67 F. Hoffman-La Roche Ltd: Financial Overview (2021)
- Figure 68 Qiagen N.V.: Financial Overview (2021)
- Figure 69 Siemens Healthineers AG (A Subsidiary of Siemens AG): Financial Overview (2022)
- Figure 70 Thermo Fisher Scientific Inc.: Financial Overview (2021)
- Figure 71 Bio-Rad Laboratories: Financial Overview (2021)
- Figure 72 Illumina, Inc.: Financial Overview (2021)
- Figure 73 Shenzhen Mindray Bio-Medical Electronics Co., Ltd: Financial Overview (2021)
- Figure 74 QuidelOrtho Corporation; Financial Overview (2021)
- Figure 75 Agilent Technologies, Inc.: Financial Overview (2022)
- Figure 76 DiaSorin S.p.A: Financial Overview (2021)
In-Vitro Diagnostics Market by Offering (Kits, Software), Technology (Immunoassay, Molecular Diagnostics [PCR, NGS, Microarray], Rapid Tests, Biochemistry), Application (Infectious Diseases, Oncology), Diagnostic Approach (Lab, POC)-Global Forecast to 2030
The global in vitro diagnostics market is expected to register a CAGR of 4.6% from 2023 to 2030 to reach $130.1 billion by 2030.
The growth of the global in vitro diagnostics market is mainly attributed to the rising prevalence of chronic diseases coupled with the increasing geriatric population, rising prevalence of acute and chronic infectious diseases, increasing funding for research activities, growing awareness regarding early disease diagnosis, growing demand for point-of-care (POC) diagnostics and rapid diagnosis, rising healthcare expenditure, and increasing funding for research activities. Moreover, the inclination of emerging economies toward personalized medicine and advancements in genomics & proteomics offer significant growth opportunities for the players operating in this market.
Based on offering, the reagents & kits segment is expected to grow with the fastest growth during the forecast period owing to its advantages as it can detect several markers simultaneously, which is useful for detecting complex diseases. Its technological superiority and the ability to facilitate chemical reactions further support the growth of this segment.
Based on the technology, in 2023, the molecular diagnostics segment is expected to account for the largest share of the market. The largest share of the segment attributed to its improved specificity, cost-savings and enhanced throughput when automated, minimizing the false positive test results by targeting specific molecules, as well as improved diagnosis of infections such as orthopedic infections.
Based on diagnostic approach, the laboratory testing segment is expected to account for the largest share of the market. Lab testing is a traditional approach in which the clinician takes a sample from the patient and sends it to the laboratory for processing and testing. Factors such as low costs of tests, highly sensitive analyzers, more accuracy and reliability, and accessibility & availability of multiple IVD tests support the largest share of the segment.
Based on application, in 2023, the infectious diseases segment is expected to account for the largest share of the global in vitro diagnostics (IVD) market owing to the high prevalence of infectious diseases, government initiatives to promote awareness and testing, and emerging outbreaks of infectious diseases around the world.
Based on end user, the hospitals & clinics segment is expected to grow with the highest CAGR during the forecast period. Patient inflows at hospitals & clinics and subsequent increase in the volume of testing carried out in hospitals and clinics due to the outbreak of COVID-19 support the growth of this segment.
An in-depth analysis of the geographical scenario of the global in-vitro diagnostics (IVD) market provides detailed qualitative and quantitative insights about the five major geographies (North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa) along with the coverage of major countries in each region. In 2023, North America is estimated to account for the largest share of the IVD market, followed by Europe, Asia-Pacific, Latin America, and Middle East & Africa. The high prevalence of chronic and infectious diseases, high awareness regarding early disease diagnosis, high adoption of advanced diagnostic products, and presence of funding coupled with novel developments in diagnostic technologies support the large share of this market.
The key players operating in the global in-vitro diagnostics market are Abbott Laboratories (U.S.), Becton, Dickinson and Company (U.S.), bioMerieux SA (France), Bio-Rad Laboratories, Inc. (U.S.), Danaher Corporation (U.S.), F. Hoffmann-La Roche Ltd. (Switzerland), Illumina, Inc. (U.S.), QIAGEN N.V. (Netherlands), Shenzhen Mindray Bio-Medical Electronics Co., Ltd (China), Siemens Healthineers AG (Germany), Thermo Fisher Scientific Inc. (U.S.), Wama Diagnostica (Brazil), Wiener Laboratorios SAIC (Argentina), QuidelOrtho Corporation (U.S.), Agilent Technologies Inc. (U.S.), and DiaSorin S.p.A. (Italy).
Scope of the Report:
Global In Vitro Diagnostics Market Assessment-by Offering
- Reagents & Kits
- Instruments
- Software & Services
Global In Vitro Diagnostics Market Assessment-by Technology
- Molecular Diagnostics
- Polymerase Chain Reaction (PCR)
- Hybridization
- Isothermal Nucleic Acid Amplification Technology
- DNA Sequencing & Next-generation Sequencing
- Microarrays
- Mass Spectrometry
- Other Molecular Diagnostic Technologies
(Other Molecular Diagnostic Technologies majorly include electrophoresis, Ligase Chain Reaction (LCR), Northern Blot, and Southern Blot)
- Point of Care (POC) Diagnostics
- Lateral Flow Assays/Rapid Tests
- POC Molecular Diagnostics
- Other PoC Products
- Immunoassay/Immunochemistry
- Enzyme-linked Immunosorbent Assays (ELISA)
- Enzyme-linked Immunospot Assays (ELISPOT)
- Western Blotting
- Radioimmunoassay
- Biochemistry/Clinical Chemistry
- Metabolic Panel
- Electrolyte Panel
- Liver Panel
- Lipid Profile
- Renal Profile
- Thyroid Function Panel
- Whole Blood Glucose Monitoring
- Microbiology
- Hematology
- Coagulation/Hemostasis
- Urinalysis
- Other IVD Technologies
(Other IVD technologies comprise anatomical pathology, histochemistry, and loop-mediated amplification).
Global In Vitro Diagnostics Market Assessment-by Application
- Infectious Diseases
- COVID-19 Testing
- Sexually Transmitted Diseases (STD) Testing
- Healthcare-associated Infections (HAIs)
- Hepatitis
- HIV
- Tropical Diseases
- Respiratory Infections (Excluding Influenza)
- Influenza
- Other Infectious Diseases
- Oncology
- Diabetes
- Cardiology
- Nephrology
- Autoimmune Disorders
- Other Applications
(Other applications comprise drugs of abuse tests, coagulation tests, neonatal testing, genetic testing, and neurological disorders testing).
(Other infectious diseases include congenital infections, meningitis, norovirus (stomach flu), hand, foot, and mouth disease (HFMD), and pertussis).
Global In Vitro Diagnostics Market Assessment-by Diagnostic Approach
- Laboratory Testing
- OTC/Self-testing
- Point-of-Care Testing
Global In Vitro Diagnostics Market Assessment-by End User
- Diagnostic Laboratories
- Hospitals & Clinics
- Home Healthcare
- Other End Users
(Other end users comprise nursing homes, academic & research institutes, ambulatory care centers, and transfusion laboratories.)
Global In vitro Diagnostics Market Assessment-by Geography
- North America
- U.S.
- Canada
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Switzerland
- Rest of Europe
- Asia-Pacific
- Japan
- China
- India
- South Korea
- Australia
- Rest of Asia-Pacific
- Latin America
- Brazil
- Mexico
- Argentina
- Rest of Latin America
- Middle East
- Saudi Arabia
- Rest of Middle East
- Africa
- South Africa
- Rest of Africa
TABLE OF CONTENTS
1. Overview
- 1.1. Market Definition & Scope
- 1.2. Market Ecosystem
- 1.3. Currency & Limitation
- 1.4. Key Stakeholders
2. Research Methodology
- 2.1. Research Process
- 2.2. Process of Data Collection & Validation
- 2.2.1. Secondary Research
- 2.2.2. Primary Research/Interviews With Key Opinion Leaders of the Industry
- 2.3. Market Sizing and Forecast
- 2.3.1. Market Size Estimation Approach
- 2.3.2. Market Share Analysis
- 2.3.3. Growth Forecast Approach
- 2.4. Assumptions for the Study
3. Executive Summary
4. Market Insights
- 4.1. Overview
- 4.2. Drivers
- 4.2.1. Rising Prevalence of Chronic Diseases Coupled With the Increasing Geriatric Population
- 4.2.2. Rising Prevalence of Infectious Diseases
- 4.2.3. Growing Demand for Point-of-Care (PoC) and Rapid Diagnostics
- 4.2.4. Rising Awareness Regarding Early Disease Diagnosis
- 4.2.5. Rising Healthcare Expenditures
- 4.2.6. Increasing Funding for Research Activities
- 4.3. Restraints
- 4.3.1. Stringent Technical Requirements and Regulatory Processes for High/Moderate-Complexity Tests
- 4.3.2. Variance in Test Results Observed in Rapid IVD Tests
- 4.4. Opportunities
- 4.4.1. Emerging Economies
- 4.4.2. Increasing Inclination Toward Personalized Medicine
- 4.4.3. Advancements in Genomics and Proteomics
- 4.5. Challenges
- 4.5.1. Evolving Regulatory Landscape
- 4.6. Technology Trends
- 4.6.1. Integration of Artificial Intelligence & Machine Learning in Molecular Diagnostics
- 4.6.2. Development of Smartphone-based Detection Platforms for Rapid Diagnostic Tests
- 4.6.3. Increasing Applications of Next-generation Sequencing Technology
5. In Vitro Diagnostics (IVD) Market: Regulatory Analysis
- 5.1. Overview
- 5.2. North America
- 5.2.1. U.S.
- 5.2.2. Canada
- 5.3. Europe
- 5.4. Asia-Pacific
- 5.4.1. China
- 5.4.2. Japan
- 5.4.3. India
- 5.5. Latin America
- 5.6. Middle East
6. In Vitro Diagnostics (IVD) Market Assessment-by Offering
- 6.1. Overview
- 6.2. Reagents & Kits
- 6.3. Systems
- 6.4. Software & Services
7. In Vitro Diagnostics (IVD) Market Assessment-by Technology
- 7.1. Overview
- 7.2. Molecular Diagnostics
- 7.2.1. Polymerase Chain Reaction (PCR)
- 7.2.2. Hybridization
- 7.2.3. Isothermal Nucleic Acid Amplification Technology
- 7.2.4. DNA Sequencing & Next-Generation Sequencing
- 7.2.5. Microarrays
- 7.2.6. Mass Spectrometry
- 7.2.7. Other Molecular Diagnostic Technologies
- 7.3. Point-of-Care (PoC) Diagnostics
- 7.3.1. Lateral Flow Assays/Rapid Tests
- 7.3.2. PoC Molecular Diagnostics
- 7.3.3. Other PoC Products
- 7.4. Immunoassay/Immunochemistry
- 7.4.1. Enzyme-linked Immunosorbent Assays (ELISA)
- 7.4.2. Enzyme-linked Immunospot Assays (ELISPOT)
- 7.4.3. Western Blotting
- 7.4.4. Radioimmunoassay
- 7.5. Biochemistry/Clinical Chemistry
- 7.5.1. Metabolic Panels
- 7.5.2. Electrolyte Panels
- 7.5.3. Liver Panels
- 7.5.4. Lipid Profiles
- 7.5.5. Renal Profiles
- 7.5.6. Thyroid Function Panels
- 7.6. Whole Blood Glucose Monitoring
- 7.7. Microbiology
- 7.8. Hematology
- 7.9. Coagulation/Hemostasis
- 7.10. Urinalysis
- 7.11. Other IVD Technologies
8. In Vitro Diagnostic (IVD) Market-by Application
- 8.1. Overview
- 8.2. Infectious Diseases
- 8.2.1. COVID-19 Testing
- 8.2.2. Sexually Transmitted Diseases (STD) Testing
- 8.2.3. Healthcare-associated Infections (HAIs)
- 8.2.4. Hepatitis
- 8.2.5. HIV
- 8.2.6. Tropical Diseases
- 8.2.7. Influenza
- 8.2.8. Respiratory Infections (Excluding Influenza)
- 8.2.9. Other Infectious Diseases
- 8.3. Oncology
- 8.4. Diabetes
- 8.5. Cardiology
- 8.6. Nephrology
- 8.7. Autoimmune Disorders
- 8.8. Other Applications
9. In Vitro Diagnostics (IVD) Market Assessment-by Diagnostic Approach
- 9.1. Overview
- 9.2. Laboratory Testing
- 9.3. OTC/Self Testing
- 9.4. Point-of-Care Testing
10. In Vitro Diagnostics (IVD) Market-by End User
- 10.1. Overview
- 10.2. Hospitals & Clinics
- 10.3. Diagnostic Laboratories
- 10.4. Home Healthcare
- 10.5. Other End Users
11. In Vitro Diagnostics (IVD) Market Assessment, by Geography
- 11.1. Overview
- 11.2. North America
- 11.2.1. U.S.
- 11.2.2. Canada
- 11.3. Europe
- 11.3.1. Germany
- 11.3.2. France
- 11.3.3. U.K.
- 11.3.4. Italy
- 11.3.5. Spain
- 11.3.6. Switzerland
- 11.3.7. Rest of Europe
- 11.4. Asia-Pacific
- 11.4.1. Japan
- 11.4.2. China
- 11.4.3. India
- 11.4.4. South Korea
- 11.4.5. Australia
- 11.4.6. Rest of Asia-Pacific
- 11.5. Latin America
- 11.5.1. Brazil
- 11.5.2. Mexico
- 11.5.3. Argentina
- 11.5.4. Rest of Latin America
- 11.6. Middle East
- 11.6.1. Saudi Arabia
- 11.6.2. Rest of Middle East
- 11.7. Africa
- 11.7.1. South Africa
- 11.7.2. Rest of Africa
12. Competitive Landscape
- 12.1. Overview
- 12.2. Key Growth Strategies
- 12.3. Competitive Dashboard
- 12.3.1. Industry Leaders
- 12.3.2. Market Differentiators
- 12.3.3. Vanguards
- 12.3.4. Emerging Companies
- 12.4. Vendor Market Positioning
- 12.5. Market Share Analysis (2022: Top 15 Players)
- 12.5.1. F. Hoffman-La Roche Ltd. (Switzerland)
- 12.5.2. Abbott Laboratories (U.S.)
- 12.5.3. Danaher Corporation (U.S)
- 12.5.4. Siemens Healthineers AG (Germany)
- 12.5.5. Thermo Fisher Scientific Inc. (U.S.)
13. Company Profiles
- 13.1. Abbott Laboratories
- 13.2. Becton, Dickinson and Company
- 13.3. biomerieux S.A.
- 13.4. Danaher Corporation
- 13.5. F. Hoffman-La Roche Ltd
- 13.6. Qiagen N.V.
- 13.7. Siemens Healthineers AG (A Subsidiary of Siemens AG)
- 13.8. Thermo Fisher Scientific Inc.
- 13.9. Bio-Rad Laboratories, Inc.
- 13.10. Wama Diagnostica
- 13.11. lllumina, Inc.
- 13.12. Wiener Laboratories SAIC
- 13.13. Shenzhen Mindray Bio-Medical Electronics Co., Ltd
- 13.14. QuidelOrtho Corporation
- 13.15. Agilent Technologies, Inc.
- 13.16. DiaSorin S.p.A.
14. Appendix
- 14.1. Available Customization
- 14.2. Related Reports