|
|
市場調査レポート
商品コード
1099254
患者モニタリング装置の世界市場Patient Monitoring Devices: Global Markets |
||||||
| 患者モニタリング装置の世界市場 |
|
出版日: 2022年07月06日
発行: BCC Research
ページ情報: 英文 204 Pages
納期: 即納可能
|
- 全表示
- 概要
- 図表
- 目次
世界の患者モニタリング装置の市場規模は、2022年に427億米ドルに達するとみられています。
同市場は、2022年~2027年の予測期間中に8.9%のCAGRで拡大し、2027年には654億米ドルに達すると予測されています。
当レポートでは、世界の患者モニタリング装置市場について調査し、市場の概要とともに、装置の接続性別、製品タイプ別、エンドユーザー業界別、地域別の動向、および市場に参入する企業のプロファイルなどを提供しています。
目次
第1章 イントロダクション
第2章 まとめとハイライト
第3章 市場概要と技術の背景
第4章 市場内訳、装置の接続性別
- 概要
- 有線接続
- 無線接続
第5章 市場内訳、製品タイプ別
- イントロダクション
- 機器市場
- マルチパラメータ患者モニター
- 血圧モニタリングデバイス
- 心臓モニター
- 呼吸器および睡眠障害モニター
- ニューロモニター
- 血糖モニタリングデバイス
- 継続的血糖モニタリングデバイス
- リモート患者モニタリングデバイス
- 温度計
- 麻酔モニター
- 血液ガス分析装置/モニター
- 胎児モニター
- 新生児モニター
- サービス
- 消耗品とアクセサリー
第6章 市場内訳、エンドユーザー別
- 概要
- 病院
- 在宅ケア
- 診療所、クリニック
- 診断センター、研究所
- プライマリヘルスケアおよびコミュニティヘルスケアセンター
- その他
第7章 市場内訳、地域別
- イントロダクション
- 北米
- 欧州
- アジア太平洋
- その他の地域
第8章 特許分析
第9章 競合情勢
第10章 企業プロファイル
- ABBOTT LABORATORIES
- A&D CO. LTD.
- AEROTEL MEDICAL SYSTEMS
- AMBU
- AMERICAN DIAGNOSTIC CORP.(ADC)
- ANALOG DEVICES INC.
- ATCOR MEDICAL PTY LTD.
- BAYER HEALTHCARE AG
- BIONET
- BIOTRONIK SE & CO. KG
- BOSTON SCIENTIFIC CORP.
- BRAUN MELSUNGEN AG
- BRIGGS HEALTHCARE
- CARDIAC SCIENCE CORP.
- CARDIOCOM LLC(MEDTRONIC)
- CARDIONET INC.
- CONMED INC.
- CONTEC MEDICAL SYSTEMS CO. LTD.
- DRAEGER
- EDWARDS LIFESCIENCES CORP.
- GE HEALTHCARE
- HONEYWELL INTERNATIONAL INC.
- INSULET CORP./OMNIPOD
- MASIMO CORP.
- MEDTRONIC
- MEMSCAP INC.
- NATUS MEDICAL INC.
- NIHON KOHDEN CORP.
- NONIN MEDICAL
- NOVA BIOMEDICAL CORP.
- OMRON CORP.(OMRON)
- OSI SYSTEMS INC.(SPACELABS HEALTHCARE)
- PHILIPS HEALTHCARE
- RADIOMETER MEDICAL APS
- ROCHE DIAGNOSTICS
- SCHILLER AG
- SIEMENS HEALTHCARE INC.(U.S.)
- SPACELABS HEALTHCARE INC.
- USCOM LTD.
- WELCH ALLYN(HILL-ROM)
- ZYNEX INC.
第11章 付録:略語のリスト
List of Tables
- Summary Table A : Global Patient Monitoring Device Market, by Region, Through 2027
- Summary Table B : Global Patient Monitoring Device Market, by Product Type, Through 2027
- Table 1 : Key Market Trends
- Table 2 : Porter's Five Forces Analysis: Overview
- Table 3 : Global Patient Monitoring Device Market, by Device Connectivity, Through 2027
- Table 4 : Global Wire-Connected Patient Monitoring Device Market, by Region, Through 2027
- Table 5 : Global Wireless-Connected Patient Monitoring Device Market, by Region, Through 2027
- Table 6 : Global Patient Monitoring Device Market, by Product Type, Through 2027
- Table 7 : Global Patient Monitoring Equipment Market, by Equipment/Device Category, Through 2027
- Table 8 : Global Multi-Parameter Patient Monitoring Device Market, by Region, Through 2027
- Table 9 : Global Blood Pressure Monitoring Device Market, by Region, Through 2027
- Table 10 : Global Cardiac Monitoring Device Market, by Region, Through 2027
- Table 11 : Global Respiratory/Sleep Disorder Monitoring Device Market, by Region, Through 2027
- Table 12 : Global Neuromonitor Market, by Region, Through 2027
- Table 13 : Global Blood Glucose Monitoring Device Market, by Region, Through 2027
- Table 14 : Global Remote Patient Monitoring Device Market, by Region, Through 2027
- Table 15 : Global Thermometer Market, by Region, Through 2027
- Table 16 : Global Anesthesia Monitoring Device Market, by Region, Through 2027
- Table 17 : Global Blood Gas Analyzer Market, by Region, Through 2027
- Table 18 : Global Fetal Monitoring Device Market, by Region, Through 2027
- Table 19 : Global Neonatal Monitoring Device Market, by Region, Through 2027
- Table 20 : Global Patient Monitoring Service Market, by Region, Through 2027
- Table 21 : Global Patient Monitoring Supply and Accessory Market, by Region, Through 2027
- Table 22 : Global Patient Monitoring Device Market, by End User, Through 2027
- Table 23 : Global Patient Monitoring Device Market, Hospital End Users, by Region, Through 2027
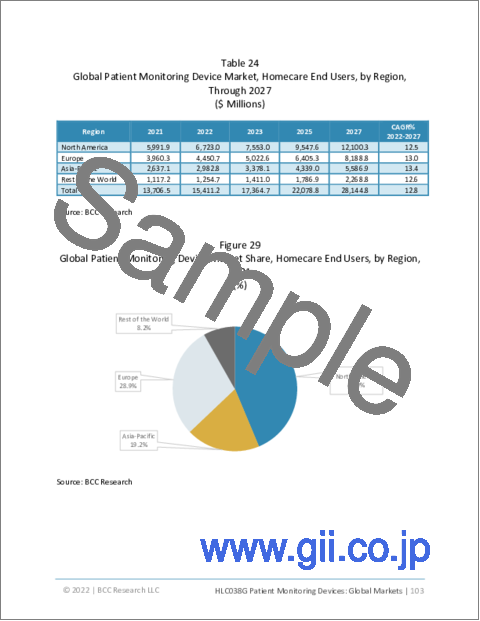
- Table 24 : Global Patient Monitoring Device Market, Homecare End Users, by Region, Through 2027
- Table 25 : Global Patient Monitoring Device Market, Medical Practitioner and Clinic End Users, by Region, Through 2027
- Table 26 : Global Patient Monitoring Device Market, Diagnostic Center and Laboratory End Users, by Region, Through 2027
- Table 27 : Global Patient Monitoring Device Market, Primary Healthcare and Community Healthcare Center End Users, by Region, Through 2027
- Table 28 : Global Patient Monitoring Device Market, Other End Users, by Region, Through 2027
- Table 29 : Global Patient Monitoring Device Market, by Region, Through 2027
- Table 30 : North American Patient Monitoring Device Market, by Device Connectivity, Through 2027
- Table 31 : North American Patient Monitoring Device Market, by Product Type, Through 2027
- Table 32 : North American Patient Monitoring Equipment Market, by Equipment/Device Category, Through 2027
- Table 33 : North American Patient Monitoring Device Market, by End User, Through 2027
- Table 34 : European Patient Monitoring Device Market, by Device Connectivity, Through 2027
- Table 35 : European Patient Monitoring Device Market, by Product Type, Through 2027
- Table 36 : European Patient Monitoring Equipment Market, by Equipment/Device Category, Through 2027
- Table 37 : European Patient Monitoring Device Market, by End User, Through 2027
- Table 38 : Asia-Pacific Patient Monitoring Device Market, by Device Connectivity, Through 2027
- Table 39 : Asia-Pacific Patient Monitoring Device Market, by Product Type, Through 2027
- Table 40 : Asia-Pacific Patient Monitoring Equipment Market, by Equipment/Device Category, Through 2027
- Table 41 : Asia-Pacific Patient Monitoring Device Market, by End User, Through 2027
- Table 42 : Rest of World Patient Monitoring Device Market, by Device Connectivity, Through 2027
- Table 43 : Rest of World Patient Monitoring Device Market, by Product Type, Through 2027
- Table 44 : Rest of World Patient Monitoring Equipment Market, by Equipment/Device Category, Through 2027
- Table 45 : Rest of World Patient Monitoring Device Market, by End User, Through 2027
- Table 46 : U.S. Patents, Patient Monitoring Devices, 2021-2022
- Table 47 : European Patents, Patient Monitoring Devices, 2020-2022
- Table 48 : Japanese Patents, Patient Monitoring Devices, 2020-2022
- Table 49 : Leading Players, Patient Monitoring Device Segments
- Table 50 : Abbott Laboratories: Recent Financial Performance, FY 2019-FY 2021
- Table 51 : Abbott Laboratories: Product Portfolio
- Table 52 : Abbott Laboratories: Recent Developments, 2022
- Table 53 : Bayer: Recent Financial Performance, FY 2019-FY 2021
- Table 54 : Bayer Healthcare: Product Portfolio
- Table 55 : Draeger AG: Recent Financial Performance, FY 2019-FY 2021
- Table 56 : Draeger AG: Product Portfolio
- Table 57 : Draeger AG: Patient Monitoring Device Product Portfolio
- Table 58 : GE Healthcare: Recent Financial Performance, FY 2019-FY 2021
- Table 59 : GE Healthcare: Product Portfolio
- Table 60 : GE Healthcare: Patient Monitoring Device Portfolio
- Table 61 : Medtronic: Recent Financial Performance, FY 2019-FY 2021
- Table 62 : Medtronic: Product Portfolio
- Table 63 : Medtronic: Patient Monitoring Device Product Portfolio
- Table 64 : Medtronic: Recent Developments, 2022
- Table 65 : Nihon Kohden Corp.: Recent Financial Performance, FY 2019-FY 2021
- Table 66 : Omron Corp.: Recent Financial Performance, FY 2019-FY 2021
- Table 67 : Omron Corp.: Patient Monitoring Device Product Portfolio
- Table 68 : Philips: Recent Financial Performance, FY 2019-FY 2021
- Table 69 : Philips Healthcare: Product Portfolio
- Table 70 : Philips Healthcare: Patient Monitoring Device Product Portfolio
- Table 71 : Roche Diagnostics: Recent Financial Performance, FY 2019-FY 2021
- Table 72 : Roche Diagnostics: Product Portfolio
- Table 73 : Roche Diagnostics: Patient Monitoring Device Product Portfolio
- Table 74 : Spacelabs Healthcare: Patient Monitoring Device Product Portfolio
- Table 75 : Welch Allyn: Patient Monitoring Device Product Portfolio
- Table 76 : Abbreviations Used in This Report
List of Figures
- Summary Figure A : Global Patient Monitoring Device Market Share, by Region, 2021
- Summary Figure B : Global Patient Monitoring Device Market Share, by Product Type, 2021
- Figure 1 : Value Chain of the Patient Monitoring Device Market
- Figure 2 : Patient Monitoring Device Market: Porter's Five Forces Analysis
- Figure 3 : Patient Monitoring Device Market: Bargaining Power of Suppliers
- Figure 4 : Patient Monitoring Device Market: Bargaining Power of Consumers
- Figure 5 : Patient Monitoring Device Market: Threat of New Entrants
- Figure 6 : Patient Monitoring Devices Market: Competitive Rivalry
- Figure 7 : Global Patient Monitoring Device Market Share, by Device Connectivity, 2021
- Figure 8 : Global Wire-Connected Patient Monitoring Device Market Share, by Region, 2021
- Figure 9 : Global Wireless-Connected Patient Monitoring Device Market Share, by Region, 2021
- Figure 10 : Global Patient Monitoring Device Market Share, by Product Type, 2021
- Figure 11 : Global Patient Monitoring Equipment Market Share, by Equipment/Device Category, 2021
- Figure 12 : Global Multi-Parameter Patient Monitoring Device Market Share, by Region, 2021
- Figure 13 : Global Blood Pressure Monitoring Device Market Share, by Region, 2021
- Figure 14 : Global Cardiac Monitoring Device Market Share, by Region, 2021
- Figure 15 : Global Respiratory/Sleep Disorder Monitoring Device Market Share, by Region, 2021
- Figure 16 : Global Neuromonitor Market Share, by Region, 2021
- Figure 17 : Global Blood Glucose Monitoring Device Market Share, by Region, 2021
- Figure 18 : Global Remote Patient Monitoring Device Market Share, by Region, 2021
- Figure 19 : Global Thermometer Market Share, by Region, 2021
- Figure 20 : Global Anesthesia Monitoring Device Market Share, by Region, 2021
- Figure 21 : Global Blood Gas Analyzer Market Share, by Region, 2021
- Figure 22 : Global Fetal Monitoring Device Market Share, by Region, 2021
- Figure 23 : Global Neonatal Monitoring Device Market Share, by Region, 2021
- Figure 24 : Global Patient Monitoring Service Market Share, by Region, 2021
- Figure 25 : Global Patient Monitoring Supply and Accessory Market Share, by Region, 2021
- Figure 26 : Global Patient Monitoring Device Market Share, by End User, 2021
- Figure 27 : Global Patient Monitoring Device Market Share, Hospital End Users, by Department, 2021
- Figure 28 : Global Patient Monitoring Device Market Share, Hospital End Users, by Region, 2021
- Figure 29 : Global Patient Monitoring Device Market Share, Homecare End Users, by Region, 2021
- Figure 30 : Global Patient Monitoring Device Market Share, Medical Practitioner and Clinic End Users, by Region, 2021
- Figure 31 : Global Patient Monitoring Device Market Share, Diagnostic Center and Laboratory End Users, by Region, 2021
- Figure 32 : Global Patient Monitoring Device Market Share, Primary Healthcare and Community Healthcare Center End Users, by Region, 2021
- Figure 33 : Global Patient Monitoring Device Market Share, Other End Users, by Region, 2021
- Figure 34 : Global Patient Monitoring Device Market Share, by Region, 2021
- Figure 35 : North American Patient Monitoring Device Market Share, by Device Connectivity, 2021
- Figure 36 : North American Patient Monitoring Device Market Share, by Product Type, 2021
- Figure 37 : North American Patient Monitoring Equipment Market Share, by Equipment/Device Category, 2021
- Figure 38 : North American Patient Monitoring Device Market Share, by End User, 2021
- Figure 39 : United States Patient Monitoring Device Market, 2021-2027
- Figure 40 : Canadian Patient Monitoring Device Market, 2021-2027
- Figure 41 : European Patient Monitoring Device Market Share, by Device Connectivity, 2021
- Figure 42 : European Patient Monitoring Device Market Share, by Product Type, 2021
- Figure 43 : European Patient Monitoring Equipment Market Share, by Equipment/Device Category, 2021
- Figure 44 : European Patient Monitoring Device Market Share, by End User, 2021
- Figure 45 : German Patient Monitoring Device Market, 2021-2027
- Figure 46 : French Patient Monitoring Device Market, 2021-2027
- Figure 47 : United Kingdom Patient Monitoring Device Market, 2021-2027
- Figure 48 : Italian Patient Monitoring Device Market, 2021-2027
- Figure 49 : Spanish Healthcare Spending Forecast, 2020-2025*
- Figure 50 : Spanish Patient Monitoring Device Market, 2021-2027
- Figure 51 : Rest of the European Patient Monitoring Device Market, 2021-2027
- Figure 52 : Asia-Pacific Patient Monitoring Device Market Share, by Device Connectivity, 2021
- Figure 53 : Asia-Pacific Patient Monitoring Device Market Share, by Product Type, 2021
- Figure 54 : Asia-Pacific Patient Monitoring Equipment Market Share, by Equipment/Device Category, 2021
- Figure 55 : Asia-Pacific Patient Monitoring Device Market Share, by End User, 2021
- Figure 56 : Japanese Patient Monitoring Device Market, 2021-2027
- Figure 57 : Chinese Patient Monitoring Device Market, 2021-2027
- Figure 58 : Indian Patient Monitoring Device Market, 2021-2027
- Figure 59 : South Korean Patient Monitoring Device Market, 2021-2027
- Figure 60 : Rest of Asia-Pacific Patient Monitoring Device Market, 2021-2027
- Figure 61 : Rest of World Patient Monitoring Device Market Share, by Device Connectivity, 2021
- Figure 62 : Rest of World Patient Monitoring Device Market Share, by Product Type, 2021
- Figure 63 : Rest of World Patient Monitoring Equipment Market Share, by Equipment/Device Category, 2021
- Figure 64 : Rest of World Patient Monitoring Device Market Share, by End User, 2021
- Figure 65 : Global Market Share, Major Players, Patient Monitoring Devices, 2021
- Figure 66 : Global Market Share, Major Players, Blood Pressure Monitors, 2021
- Figure 67 : Global Market Share, Major Players, Cardiac Monitoring Devices, 2021
- Figure 68 : Abbott Laboratories: Revenue Share, by Product Segment, FY 2021
- Figure 69 : Abbott Laboratories: Revenue Share, by Medical Device, FY 2021
- Figure 70 : Bayer: Revenue Share, by Business Segment, FY 2021
- Figure 71 : Draeger AG: Revenue Share, by Business Segment, FY 2021
- Figure 72 : GE Healthcare: Revenue Share, by Product Segment, FY 2021
- Figure 73 : Medtronic: Revenue Share, by Product Segment, FY 2021
- Figure 74 : Nihon Kohden Corp.: Revenue Share, by Product Segment, FY 2021
- Figure 75 : Nihon Kohden Corp.: Revenue Share, by Region, FY 2021
- Figure 76 : Philips: Revenue Share, by Operating Segment, FY 2021
- Figure 77 : Roche Diagnostics: Revenue Share, by Operating Segment, FY 2021
- Figure 78 : Roche Diagnostics: Revenue Share, by Region, FY 2021
Highlights:
The global patient monitoring devices market should reach $42.7 billion by 2022 and more than $65.4 billion by 2027, with a compound annual growth rate (CAGR) of 8.9% during the forecast period of 2022-2027.
The equipment segment of the global patient monitoring devices market should reach $35.4 billion by 2022 and more than $54.6 billion by 2027, with a CAGR of 9% during the forecast period of 2022-2027.
The services segment of the global patient monitoring devices market should reach $4 billion by 2022 and more than $6 billion by 2027, with a CAGR of 8.5% during the forecast period of 2022-2027.
Report Scope:
This report covers a range of products, some are disease specific and others are used for all types of patients and in general for health management. The patient monitoring device market referred to in this report is limited to the devices/products included in this report. Each product market covered in this report is unique and has a different set of market dynamics and competitive landscape. The product lifecycle of each product segment also varies.
The study will enable the audience to understand and gain insights on the current market and will forecast the changing market scenario. The data provided can help users understand which market segments among device connectivity, product type and end users are expected to grow at higher rates: factors driving growth, factors limiting the growth, key opportunity areas and so on.
Discussion and analysis cover the following market factors -
- Key market analysis.
- Market dynamics (drivers and restraints)
- Market forecast period of 2022-2027.
- Competitive developments and landscape.
- Geographic regional analysis.
- Patents.
- Profiles of key market players.
The report also covers a detailed competitive outlook, including market share and company profiles of the key participants operating in the global market. Key players profiled in the report include Abbott Laboratories, GE Healthcare, Philips Healthcare, Nihon Kohden, Omron Healthcare, Roche Diagnostics Ltd., Cardiocom, Covidien, CR Bard, Honeywell Med, Infinium Medical and InTechnology etc.
Report Includes:
- 54 data tables and 24 additional tables
- A comprehensive overview of the global market for patient monitoring devices within the healthcare industry
- Estimation of the market size and analyses of market trends, with data from 2020, 2021, estimates for 2022 and projections of compound annual growth rates (CAGRs) through 2027
- Highlights of the market potential for patient monitoring devices by product types, device categories and end users
- Identification of market drivers, restraints and other forces impacting the global market and evaluation of current market size and forecast, and assessment of new developments in the industry
- Market share analysis of the key companies of the industry and coverage of events like mergers & acquisitions, joint ventures, collaborations or partnerships, and other key market strategies and a relevant patent analysis
- Company profiles of major market players within the industry Abbott Laboratories, Bayer Healthcare AG, GE Healthcare, Medtronic, Philips Healthcare
Table of Contents
Chapter 1 Introduction
- Overview
- Study Goals and Objectives
- Scope of Report
- What's New in this Update?
- Reasons for Doing This Study
- Intended Audience
- Information Sources
- Methodology
- Geographic Breakdown
- Analyst's Credentials
- BCC Custom Research
- Related BCC Research Reports
Chapter 2 Summary and Highlights
Chapter 3 Market Overview and Technology Background
- Overview
- Patient Monitoring Device Market: Product Analysis
- Multiparameter Patient Monitors
- Blood Pressure Monitors
- Cardiac Monitoring Devices
- Other Devices
- Key Market Trends
- Market Drivers
- Expanding Healthcare Awareness and Infrastructure
- Rising Incidence of Lifestyle, Chronic Disease and Aging Populations
- Growing Popularity of Remote/Home Patient Monitoring
- Growing Trend of Acquisitions
- Continuous Product Development and Technological Advancements
- Other Factors
- Market Restraints
- Competitive Market Creating Price Pressure and Increasing Market Challenges
- Regulatory and Reimbursement Issues
- Technology and Product Adoption
- Other Restraints
- Market Opportunities
- Increasing Demand for Non-Invasive and Minimally-Invasive Devices
- Value Chain Analysis
- Porter's Five Forces Analysis
- Bargaining Power of Suppliers
- Bargaining Power of Consumers
- Threat of New Entrants
- Competitive Rivalry
- Threat of Substitutes
- Regulations and Standards Analysis
- United States
- European Union (EU)
- Japan
- Reimbursement Landscape
- Future of Patient Monitoring Devices
- Assessment of COVID-19 Impact
Chapter 4 Market Breakdown, by Device Connectivity
- Overview
- Wired Connectivity
- Wireless Connectivity
Chapter 5 Market Breakdown, by Product Type
- Introduction
- Equipment Market
- Multiparameter Patient Monitors
- Blood Pressure Monitoring Devices
- Cardiac Monitors
- Respiratory and Sleep Disorder Monitors
- Neuromonitors
- Blood Glucose Monitoring Devices
- Continuous Blood Glucose Monitoring Devices
- Remote Patient Monitoring Devices
- Thermometers
- Anesthesia Monitors
- Blood Gas Analyzers/Monitors
- Fetal Monitors
- Neonatal Monitors
- Services
- Supplies and Accessories
Chapter 6 Market Breakdown, by End-User
- Overview
- Hospitals
- Homecare
- Medical Practitioners and Clinics
- Diagnostic Centers and Laboratories
- Primary Healthcare and Community Healthcare Centers
- Others
Chapter 7 Market Breakdown, by Region
- Introduction
- North America
- United States
- Canada
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- Japan
- China
- India
- South Korea
- Rest of Asia-Pacific
- Rest of World (RoW)
Chapter 8 Patent Analysis
- Overview
- New Patent Development
- Impact on Market
Chapter 9 Competitive Landscape
- Overview
- Competitive Developments
Chapter 10 Company Profiles
- ABBOTT LABORATORIES
- A&D CO. LTD.
- AEROTEL MEDICAL SYSTEMS
- AMBU
- AMERICAN DIAGNOSTIC CORP. (ADC)
- ANALOG DEVICES INC.
- ATCOR MEDICAL PTY LTD.
- BAYER HEALTHCARE AG
- BIONET
- BIOTRONIK SE & CO. KG
- BOSTON SCIENTIFIC CORP.
- BRAUN MELSUNGEN AG
- BRIGGS HEALTHCARE
- CARDIAC SCIENCE CORP.
- CARDIOCOM LLC (MEDTRONIC)
- CARDIONET INC.
- CONMED INC.
- CONTEC MEDICAL SYSTEMS CO. LTD.
- DRAEGER
- EDWARDS LIFESCIENCES CORP.
- GE HEALTHCARE
- HONEYWELL INTERNATIONAL INC.
- INSULET CORP./OMNIPOD
- MASIMO CORP.
- MEDTRONIC
- MEMSCAP INC.
- NATUS MEDICAL INC.
- NIHON KOHDEN CORP.
- NONIN MEDICAL
- NOVA BIOMEDICAL CORP.
- OMRON CORP. (OMRON)
- OSI SYSTEMS INC. (SPACELABS HEALTHCARE)
- PHILIPS HEALTHCARE
- RADIOMETER MEDICAL APS
- ROCHE DIAGNOSTICS
- SCHILLER AG
- SIEMENS HEALTHCARE INC. (U.S.)
- SPACELABS HEALTHCARE INC.
- USCOM LTD.
- WELCH ALLYN (HILL-ROM)
- ZYNEX INC.